amlodipine-besylate
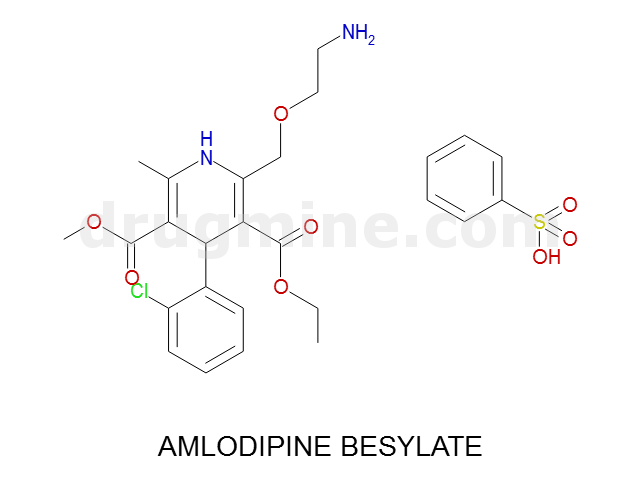

Name: AMLODIPINE BESYLATE
ID :
MW: 409
Number of atoms: 28
Molecular_Formula: C20H25ClN2O5
Alogp: 1.576
Indication class : Antihypertensive; Anti-Anginal
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
amlodipine besylate containing products summary
There are in total 243 different products containing the active ingredient amlodipine besylate. From the 243 drug products, 21 have been discontinued.Product id = 25492
Application Number = 19787
Date of Application = 31,, Jul, 1992
RX/OTC/DISCN = RX
Tradename = NORVASC
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 25493
Application Number = 19787
Date of Application = 31,, Jul, 1992
RX/OTC/DISCN = RX
Tradename = NORVASC
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 25494
Application Number = 19787
Date of Application = 31,, Jul, 1992
RX/OTC/DISCN = RX
Tradename = NORVASC
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 003
Tecode = AB
Rld = Yes
Strength = EQ 10MG BASE
----
Product id = 2403
Application Number = 20364
Date of Application = 3,, Mar, 1995
RX/OTC/DISCN = RX
Tradename = LOTREL
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 2404
Application Number = 20364
Date of Application = 3,, Mar, 1995
RX/OTC/DISCN = RX
Tradename = LOTREL
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 2405
Application Number = 20364
Date of Application = 3,, Mar, 1995
RX/OTC/DISCN = RX
Tradename = LOTREL
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 2407
Application Number = 20364
Date of Application = 20,, Jun, 2002
RX/OTC/DISCN = RX
Tradename = LOTREL
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 2408
Application Number = 20364
Date of Application = 11,, Apr, 2006
RX/OTC/DISCN = RX
Tradename = LOTREL
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 006
Tecode = AB
Rld = Yes
Strength = EQ 10MG BASE;40MG
----
Product id = 2406
Application Number = 20364
Date of Application = 11,, Apr, 2006
RX/OTC/DISCN = RX
Tradename = LOTREL
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 007
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 18775
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 10MG BASE
----
Product id = 18776
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 20MG BASE
----
Product id = 18777
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 40MG BASE
----
Product id = 18778
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 80MG BASE
----
Product id = 18779
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 10MG BASE
----
Product id = 18780
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 20MG BASE
----
Product id = 18781
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 007
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 40MG BASE
----
Product id = 18782
Application Number = 21540
Date of Application = 30,, Jan, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 008
Tecode = AB
Rld = Yes
Strength = EQ 10MG BASE;EQ 80MG BASE
----
Product id = 18772
Application Number = 21540
Date of Application = 29,, Jul, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 009
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 10MG BASE
----
Product id = 18773
Application Number = 21540
Date of Application = 29,, Jul, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 010
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 20MG BASE
----
Product id = 18774
Application Number = 21540
Date of Application = 29,, Jul, 2004
RX/OTC/DISCN = RX
Tradename = CADUET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = PFIZER
ProductNo = 011
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 40MG BASE
----
Product id = 21128
Application Number = 21990
Date of Application = 20,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = EXFORGE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;160MG
----
Product id = 21130
Application Number = 21990
Date of Application = 20,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = EXFORGE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 003
Tecode = AB
Rld = Yes
Strength = EQ 10MG BASE;160MG
----
Product id = 21129
Application Number = 21990
Date of Application = 20,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = EXFORGE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;320MG
----
Product id = 21131
Application Number = 21990
Date of Application = 20,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = EXFORGE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 005
Tecode = AB
Rld = Yes
Strength = EQ 10MG BASE;320MG
----
Product id = 16847
Application Number = 22026
Date of Application = 27,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET, ORALLY DISINTEGRATING
Application Type = N
Applicant Name = SYNTHON PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 16848
Application Number = 22026
Date of Application = 27,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET, ORALLY DISINTEGRATING
Application Type = N
Applicant Name = SYNTHON PHARMS
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 16849
Application Number = 22026
Date of Application = 27,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET, ORALLY DISINTEGRATING
Application Type = N
Applicant Name = SYNTHON PHARMS
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 18278
Application Number = 22100
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 18279
Application Number = 22100
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 18280
Application Number = 22100
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 18281
Application Number = 22100
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 004
Tecode =
Rld = Yes
Strength = EQ 10MG BASE;40MG
----
Product id = 21132
Application Number = 22314
Date of Application = 30,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = EXFORGE HCT
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG;12.5MG;160MG
----
Product id = 21133
Application Number = 22314
Date of Application = 30,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = EXFORGE HCT
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 5MG;25MG;160MG
----
Product id = 21134
Application Number = 22314
Date of Application = 30,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = EXFORGE HCT
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 10MG;12.5MG;160MG
----
Product id = 21135
Application Number = 22314
Date of Application = 30,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = EXFORGE HCT
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG;25MG;160MG
----
Product id = 21136
Application Number = 22314
Date of Application = 30,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = EXFORGE HCT
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 005
Tecode = AB
Rld = Yes
Strength = 10MG;25MG;320MG
----
Product id = 29259
Application Number = 22401
Date of Application = 16,, Oct, 2009
RX/OTC/DISCN = RX
Tradename = TWYNSTA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 29261
Application Number = 22401
Date of Application = 16,, Oct, 2009
RX/OTC/DISCN = RX
Tradename = TWYNSTA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 29260
Application Number = 22401
Date of Application = 16,, Oct, 2009
RX/OTC/DISCN = RX
Tradename = TWYNSTA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;80MG
----
Product id = 29262
Application Number = 22401
Date of Application = 16,, Oct, 2009
RX/OTC/DISCN = RX
Tradename = TWYNSTA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 004
Tecode = AB
Rld = Yes
Strength = EQ 10MG BASE;80MG
----
Product id = 28491
Application Number = 22545
Date of Application = 26,, Aug, 2010
RX/OTC/DISCN = RX
Tradename = TEKAMLO
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 150MG BASE;EQ 5MG BASE
----
Product id = 28492
Application Number = 22545
Date of Application = 26,, Aug, 2010
RX/OTC/DISCN = RX
Tradename = TEKAMLO
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 150MG BASE;EQ 10MG BASE
----
Product id = 28493
Application Number = 22545
Date of Application = 26,, Aug, 2010
RX/OTC/DISCN = RX
Tradename = TEKAMLO
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 300MG BASE;EQ 5MG BASE
----
Product id = 28494
Application Number = 22545
Date of Application = 26,, Aug, 2010
RX/OTC/DISCN = RX
Tradename = TEKAMLO
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 004
Tecode =
Rld = Yes
Strength = EQ 300MG BASE;EQ 10MG BASE
----
Product id = 17860
Application Number = 76418
Date of Application = 3,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17861
Application Number = 76418
Date of Application = 3,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17862
Application Number = 76418
Date of Application = 3,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17831
Application Number = 76692
Date of Application = 20,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17832
Application Number = 76692
Date of Application = 20,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17833
Application Number = 76692
Date of Application = 20,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17822
Application Number = 76719
Date of Application = 23,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17823
Application Number = 76719
Date of Application = 23,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17824
Application Number = 76719
Date of Application = 23,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17886
Application Number = 76846
Date of Application = 28,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17887
Application Number = 76846
Date of Application = 28,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17888
Application Number = 76846
Date of Application = 28,, Jun, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17875
Application Number = 76859
Date of Application = 10,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17876
Application Number = 76859
Date of Application = 10,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17877
Application Number = 76859
Date of Application = 10,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17828
Application Number = 77073
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17829
Application Number = 77073
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17830
Application Number = 77073
Date of Application = 26,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17883
Application Number = 77080
Date of Application = 27,, Jun, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SYNTHON PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17884
Application Number = 77080
Date of Application = 27,, Jun, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SYNTHON PHARMS
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17885
Application Number = 77080
Date of Application = 27,, Jun, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SYNTHON PHARMS
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 1003
Application Number = 77179
Date of Application = 18,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 1004
Application Number = 77179
Date of Application = 18,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 1005
Application Number = 77179
Date of Application = 18,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 1007
Application Number = 77179
Date of Application = 18,, May, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 1006
Application Number = 77179
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 1008
Application Number = 77179
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 979
Application Number = 77183
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 980
Application Number = 77183
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 981
Application Number = 77183
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 983
Application Number = 77183
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 17872
Application Number = 77262
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17873
Application Number = 77262
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17874
Application Number = 77262
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17837
Application Number = 77333
Date of Application = 17,, Jul, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GEDEON RICHTER USA
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17838
Application Number = 77333
Date of Application = 17,, Jul, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GEDEON RICHTER USA
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17839
Application Number = 77333
Date of Application = 17,, Jul, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GEDEON RICHTER USA
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17840
Application Number = 77362
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GENPHARM
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17841
Application Number = 77362
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GENPHARM
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17842
Application Number = 77362
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GENPHARM
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 991
Application Number = 77375
Date of Application = 21,, May, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 992
Application Number = 77375
Date of Application = 21,, May, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 993
Application Number = 77375
Date of Application = 21,, May, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 995
Application Number = 77375
Date of Application = 21,, May, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 17901
Application Number = 77516
Date of Application = 11,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VIVIMED LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17902
Application Number = 77516
Date of Application = 11,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VIVIMED LABS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17903
Application Number = 77516
Date of Application = 11,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VIVIMED LABS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17904
Application Number = 77671
Date of Application = 19,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17905
Application Number = 77671
Date of Application = 19,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17906
Application Number = 77671
Date of Application = 19,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17895
Application Number = 77759
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17896
Application Number = 77759
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17897
Application Number = 77759
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17843
Application Number = 77771
Date of Application = 12,, Apr, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HIKMA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17844
Application Number = 77771
Date of Application = 12,, Apr, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HIKMA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17845
Application Number = 77771
Date of Application = 12,, Apr, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HIKMA PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 1009
Application Number = 77890
Date of Application = 14,, Oct, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 1010
Application Number = 77890
Date of Application = 14,, Oct, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 1011
Application Number = 77890
Date of Application = 14,, Oct, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 1012
Application Number = 77890
Date of Application = 14,, Oct, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 17846
Application Number = 77955
Date of Application = 28,, Aug, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17847
Application Number = 77955
Date of Application = 28,, Aug, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17848
Application Number = 77955
Date of Application = 28,, Aug, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17869
Application Number = 77974
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17870
Application Number = 77974
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17871
Application Number = 77974
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17825
Application Number = 78021
Date of Application = 17,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17826
Application Number = 78021
Date of Application = 17,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17827
Application Number = 78021
Date of Application = 17,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17849
Application Number = 78043
Date of Application = 12,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17850
Application Number = 78043
Date of Application = 12,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17851
Application Number = 78043
Date of Application = 12,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17857
Application Number = 78081
Date of Application = 31,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARMA
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17858
Application Number = 78081
Date of Application = 31,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARMA
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17859
Application Number = 78081
Date of Application = 31,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARMA
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17866
Application Number = 78131
Date of Application = 4,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PURACAP PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17867
Application Number = 78131
Date of Application = 4,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PURACAP PHARM
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17868
Application Number = 78131
Date of Application = 4,, Sep, 2007
RX/OTC/DISCN = DISCN
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PURACAP PHARM
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17854
Application Number = 78224
Date of Application = 27,, Feb, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MATRIX LABS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17855
Application Number = 78224
Date of Application = 27,, Feb, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MATRIX LABS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17856
Application Number = 78224
Date of Application = 27,, Feb, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MATRIX LABS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17910
Application Number = 78226
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17911
Application Number = 78226
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17912
Application Number = 78226
Date of Application = 9,, Jul, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17880
Application Number = 78231
Date of Application = 30,, Nov, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17881
Application Number = 78231
Date of Application = 30,, Nov, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17882
Application Number = 78231
Date of Application = 30,, Nov, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 997
Application Number = 78381
Date of Application = 29,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 998
Application Number = 78381
Date of Application = 29,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 999
Application Number = 78381
Date of Application = 29,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 1001
Application Number = 78381
Date of Application = 29,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 1000
Application Number = 78381
Date of Application = 29,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 1002
Application Number = 78381
Date of Application = 29,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 17898
Application Number = 78414
Date of Application = 7,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VINTAGE
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17899
Application Number = 78414
Date of Application = 7,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VINTAGE
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17900
Application Number = 78414
Date of Application = 7,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VINTAGE
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17863
Application Number = 78453
Date of Application = 2,, Jul, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17864
Application Number = 78453
Date of Application = 2,, Jul, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17865
Application Number = 78453
Date of Application = 2,, Jul, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 985
Application Number = 78466
Date of Application = 5,, Feb, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 986
Application Number = 78466
Date of Application = 5,, Feb, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 987
Application Number = 78466
Date of Application = 5,, Feb, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 989
Application Number = 78466
Date of Application = 5,, Feb, 2010
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN PHARMS
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 988
Application Number = 78466
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN PHARMS
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 990
Application Number = 78466
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN PHARMS
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 17819
Application Number = 78477
Date of Application = 16,, Jan, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17820
Application Number = 78477
Date of Application = 16,, Jan, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17821
Application Number = 78477
Date of Application = 16,, Jan, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17907
Application Number = 78500
Date of Application = 6,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17908
Application Number = 78500
Date of Application = 6,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17909
Application Number = 78500
Date of Application = 6,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17834
Application Number = 78552
Date of Application = 8,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = EPIC PHARMA LLC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17835
Application Number = 78552
Date of Application = 8,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = EPIC PHARMA LLC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17836
Application Number = 78552
Date of Application = 8,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = EPIC PHARMA LLC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17889
Application Number = 78573
Date of Application = 22,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17890
Application Number = 78573
Date of Application = 22,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17891
Application Number = 78573
Date of Application = 22,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17816
Application Number = 78925
Date of Application = 4,, May, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ALKEM
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17817
Application Number = 78925
Date of Application = 4,, May, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ALKEM
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17818
Application Number = 78925
Date of Application = 4,, May, 2009
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ALKEM
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 994
Application Number = 79047
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 996
Application Number = 79047
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 17935
Application Number = 90011
Date of Application = 28,, Mar, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND VALSARTAN
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;160MG
----
Product id = 17937
Application Number = 90011
Date of Application = 28,, Mar, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND VALSARTAN
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;160MG
----
Product id = 17938
Application Number = 90011
Date of Application = 28,, Mar, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND VALSARTAN
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;320MG
----
Product id = 17936
Application Number = 90144
Date of Application = 28,, Mar, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND VALSARTAN
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;320MG
----
Product id = 982
Application Number = 90149
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 984
Application Number = 90149
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 1013
Application Number = 90364
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 1014
Application Number = 90364
Date of Application = 5,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 17878
Application Number = 90752
Date of Application = 15,, Apr, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SECAN PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17879
Application Number = 90752
Date of Application = 15,, Apr, 2011
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SECAN PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 967
Application Number = 91431
Date of Application = 30,, Dec, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 968
Application Number = 91431
Date of Application = 30,, Dec, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 969
Application Number = 91431
Date of Application = 30,, Dec, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 970
Application Number = 91431
Date of Application = 30,, Dec, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 971
Application Number = 91431
Date of Application = 30,, Dec, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 972
Application Number = 91431
Date of Application = 30,, Dec, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 17992
Application Number = 200045
Date of Application = 21,, Dec, 2010
RX/OTC/DISCN = RX
Tradename = AMTURNIDE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 150MG BASE;EQ 5MG BASE;12.5MG
----
Product id = 17993
Application Number = 200045
Date of Application = 21,, Dec, 2010
RX/OTC/DISCN = RX
Tradename = AMTURNIDE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 300MG BASE;EQ 5MG BASE;12.5MG
----
Product id = 17994
Application Number = 200045
Date of Application = 21,, Dec, 2010
RX/OTC/DISCN = RX
Tradename = AMTURNIDE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 300MG BASE;EQ 5MG BASE;25MG
----
Product id = 17995
Application Number = 200045
Date of Application = 21,, Dec, 2010
RX/OTC/DISCN = RX
Tradename = AMTURNIDE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 004
Tecode =
Rld = No
Strength = EQ 300MG BASE;EQ 10MG BASE;12.5MG
----
Product id = 17996
Application Number = 200045
Date of Application = 21,, Dec, 2010
RX/OTC/DISCN = RX
Tradename = AMTURNIDE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 005
Tecode =
Rld = Yes
Strength = EQ 300MG BASE;EQ 10MG BASE;25MG
----
Product id = 29146
Application Number = 200175
Date of Application = 23,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = TRIBENZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 5MG BASE;12.5MG;20MG
----
Product id = 29147
Application Number = 200175
Date of Application = 23,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = TRIBENZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE;12.5MG;40MG
----
Product id = 29148
Application Number = 200175
Date of Application = 23,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = TRIBENZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 5MG BASE;25MG;40MG
----
Product id = 29149
Application Number = 200175
Date of Application = 23,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = TRIBENZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 004
Tecode =
Rld = No
Strength = EQ 10MG BASE;12.5MG;40MG
----
Product id = 29150
Application Number = 200175
Date of Application = 23,, Jul, 2010
RX/OTC/DISCN = RX
Tradename = TRIBENZOR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = DAIICHI SANKYO
ProductNo = 005
Tecode =
Rld = Yes
Strength = EQ 10MG BASE;25MG;40MG
----
Product id = 17924
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 10MG BASE
----
Product id = 17925
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 20MG BASE
----
Product id = 17926
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 40MG BASE
----
Product id = 17927
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 10MG BASE
----
Product id = 17928
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 20MG BASE
----
Product id = 17929
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 40MG BASE
----
Product id = 17930
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 007
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 80MG BASE
----
Product id = 17931
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 008
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 10MG BASE
----
Product id = 17932
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 009
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 20MG BASE
----
Product id = 17933
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 010
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 40MG BASE
----
Product id = 17934
Application Number = 200465
Date of Application = 29,, Nov, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 011
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 80MG BASE
----
Product id = 17852
Application Number = 201380
Date of Application = 13,, Apr, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MACLEODS PHARMS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17853
Application Number = 201380
Date of Application = 13,, Apr, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MACLEODS PHARMS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 28523
Application Number = 201586
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 28525
Application Number = 201586
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 28524
Application Number = 201586
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;80MG
----
Product id = 28526
Application Number = 201586
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = LUPIN LTD
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;80MG
----
Product id = 973
Application Number = 202239
Date of Application = 5,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;10MG
----
Product id = 974
Application Number = 202239
Date of Application = 5,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;10MG
----
Product id = 975
Application Number = 202239
Date of Application = 5,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;20MG
----
Product id = 976
Application Number = 202239
Date of Application = 5,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 977
Application Number = 202239
Date of Application = 5,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;20MG
----
Product id = 978
Application Number = 202239
Date of Application = 5,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 28527
Application Number = 202516
Date of Application = 26,, Aug, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 28529
Application Number = 202516
Date of Application = 26,, Aug, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 28528
Application Number = 202516
Date of Application = 26,, Aug, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;80MG
----
Product id = 28530
Application Number = 202516
Date of Application = 26,, Aug, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;80MG
----
Product id = 28531
Application Number = 202517
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;40MG
----
Product id = 28533
Application Number = 202517
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;40MG
----
Product id = 28532
Application Number = 202517
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;80MG
----
Product id = 28534
Application Number = 202517
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = TELMISARTAN AND AMLODIPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LTD
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;80MG
----
Product id = 17813
Application Number = 202553
Date of Application = 29,, Apr, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ACCORD HLTHCARE
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17814
Application Number = 202553
Date of Application = 29,, Apr, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ACCORD HLTHCARE
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17815
Application Number = 202553
Date of Application = 29,, Apr, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ACCORD HLTHCARE
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17892
Application Number = 203245
Date of Application = 21,, Oct, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM LABS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE
----
Product id = 17893
Application Number = 203245
Date of Application = 21,, Oct, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM LABS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 17894
Application Number = 203245
Date of Application = 21,, Oct, 2013
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM LABS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 17913
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 10MG BASE
----
Product id = 17914
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 20MG BASE
----
Product id = 17915
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 2.5MG BASE;EQ 40MG BASE
----
Product id = 17916
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 10MG BASE
----
Product id = 17917
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 005
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 20MG BASE
----
Product id = 17918
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 006
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 40MG BASE
----
Product id = 17919
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 007
Tecode = AB
Rld = No
Strength = EQ 5MG BASE;EQ 80MG BASE
----
Product id = 17920
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 008
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 10MG BASE
----
Product id = 17921
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 009
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 20MG BASE
----
Product id = 17922
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 010
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 40MG BASE
----
Product id = 17923
Application Number = 203874
Date of Application = 7,, Mar, 2014
RX/OTC/DISCN = RX
Tradename = AMLODIPINE BESYLATE AND ATORVASTATIN CALCIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 011
Tecode = AB
Rld = No
Strength = EQ 10MG BASE;EQ 80MG BASE
----
Product id = 26657
Application Number = 205003
Date of Application = 21,, Jan, 2015
RX/OTC/DISCN = RX
Tradename = PRESTALIA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SYMPLMED PHARMS LLC
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 2.5MG BASE;3.5MG
----
Product id = 26658
Application Number = 205003
Date of Application = 21,, Jan, 2015
RX/OTC/DISCN = RX
Tradename = PRESTALIA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SYMPLMED PHARMS LLC
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 5MG BASE;7MG
----
Product id = 26659
Application Number = 205003
Date of Application = 21,, Jan, 2015
RX/OTC/DISCN = RX
Tradename = PRESTALIA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SYMPLMED PHARMS LLC
ProductNo = 003
Tecode =
Rld = Yes
Strength = EQ 10MG BASE;14MG
----