betaxolol-hydrochloride

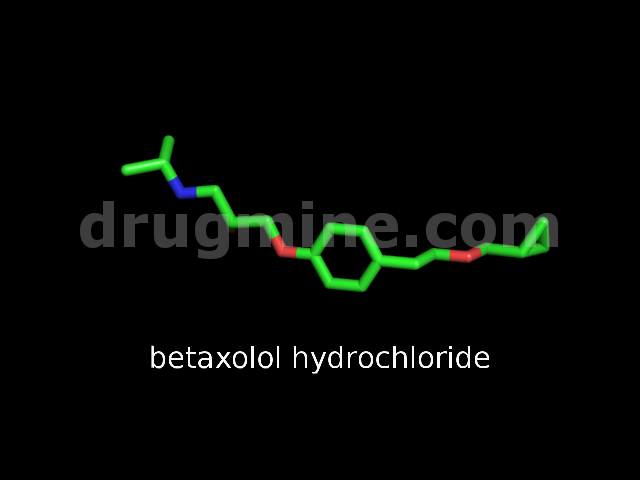
Name: BETAXOLOL HYDROCHLORIDE
ID :
MW: 307
Number of atoms: 22
Molecular_Formula: C18H29NO3
Alogp: 2.577
Indication class : Antihypertensive; Anti-Anginal
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
betaxolol hydrochloride containing products summary
There are in total 15 different products containing the active ingredient betaxolol hydrochloride. From the 15 drug products, 6 have been discontinued.Product id = 12877
Application Number = 19270
Date of Application = 30,, Aug, 1985
RX/OTC/DISCN = RX
Tradename = BETOPTIC
Route/format = OPHTHALMIC / SOLUTION/DROPS
Application Type = N
Applicant Name = ALCON
ProductNo = 001
Tecode = AT
Rld = Yes
Strength = EQ 0.5% BASE
----
Product id = 23163
Application Number = 19507
Date of Application = 27,, Oct, 1989
RX/OTC/DISCN = DISCN
Tradename = KERLONE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = No
Strength = 10MG
----
Product id = 23164
Application Number = 19507
Date of Application = 27,, Oct, 1989
RX/OTC/DISCN = DISCN
Tradename = KERLONE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 002
Tecode =
Rld = No
Strength = 20MG
----
Product id = 23161
Application Number = 19807
Date of Application = 30,, Oct, 1992
RX/OTC/DISCN = DISCN
Tradename = KERLEDEX
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG;12.5MG
----
Product id = 23162
Application Number = 19807
Date of Application = 30,, Oct, 1992
RX/OTC/DISCN = DISCN
Tradename = KERLEDEX
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG;12.5MG
----
Product id = 14543
Application Number = 19845
Date of Application = 29,, Dec, 1989
RX/OTC/DISCN = RX
Tradename = BETOPTIC S
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = ALCON
ProductNo = 001
Tecode =
Rld = Yes
Strength = EQ 0.25% BASE
----
Product id = 14542
Application Number = 20619
Date of Application = 17,, Apr, 1997
RX/OTC/DISCN = DISCN
Tradename = BETOPTIC PILO
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = ALCON
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 0.25% BASE;1.75%
----
Product id = 12871
Application Number = 75386
Date of Application = 30,, Jun, 2000
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = OPHTHALMIC / SOLUTION/DROPS
Application Type = A
Applicant Name = AKORN
ProductNo = 001
Tecode = AT
Rld = No
Strength = EQ 0.5% BASE
----
Product id = 12872
Application Number = 75446
Date of Application = 28,, Sep, 2000
RX/OTC/DISCN = DISCN
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = OPHTHALMIC / SOLUTION/DROPS
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 0.5% BASE
----
Product id = 18481
Application Number = 75541
Date of Application = 22,, Oct, 1999
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = EPIC PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 18482
Application Number = 75541
Date of Application = 22,, Oct, 1999
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = EPIC PHARMA
ProductNo = 002
Tecode = AB
Rld = Yes
Strength = 20MG
----
Product id = 12873
Application Number = 75630
Date of Application = 12,, Apr, 2001
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = OPHTHALMIC / SOLUTION/DROPS
Application Type = A
Applicant Name = IGI LABS INC
ProductNo = 001
Tecode = AT
Rld = No
Strength = EQ 0.5% BASE
----
Product id = 12874
Application Number = 78694
Date of Application = 16,, Nov, 2009
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = OPHTHALMIC / SOLUTION/DROPS
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 001
Tecode = AT
Rld = No
Strength = EQ 0.5% BASE
----
Product id = 18483
Application Number = 78962
Date of Application = 27,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = KVK TECH
ProductNo = 001
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 18484
Application Number = 78962
Date of Application = 27,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = BETAXOLOL HYDROCHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = KVK TECH
ProductNo = 002
Tecode = AB
Rld = No
Strength = 20MG
----