bumetanide
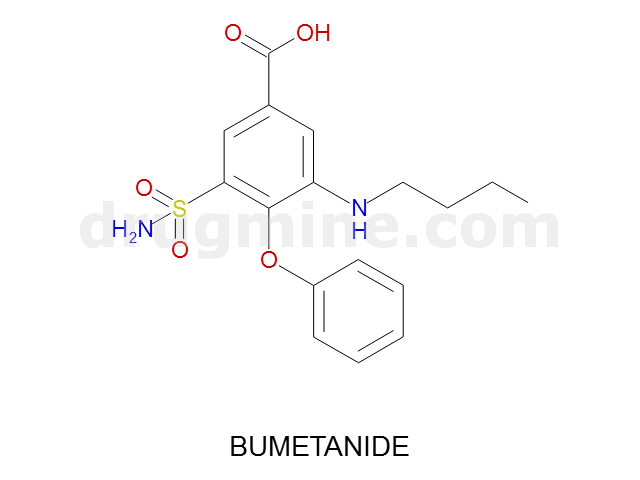

Name: BUMETANIDE
ID :
MW: 364
Number of atoms: 25
Molecular_Formula: C17H20N2O5S
Alogp: 2.862
Indication class : Diuretic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
bumetanide containing products summary
There are in total 15 different products containing the active ingredient bumetanide. From the 15 drug products, 6 have been discontinued.Product id = 18645
Application Number = 18225
Date of Application = 28,, Feb, 1983
RX/OTC/DISCN = DISCN
Tradename = BUMEX
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = VALIDUS PHARMS INC
ProductNo = 001
Tecode =
Rld = No
Strength = 1MG
----
Product id = 18644
Application Number = 18225
Date of Application = 28,, Feb, 1983
RX/OTC/DISCN = DISCN
Tradename = BUMEX
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = VALIDUS PHARMS INC
ProductNo = 002
Tecode =
Rld = No
Strength = 0.5MG
----
Product id = 18646
Application Number = 18225
Date of Application = 14,, Jun, 1985
RX/OTC/DISCN = DISCN
Tradename = BUMEX
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = VALIDUS PHARMS INC
ProductNo = 003
Tecode =
Rld = No
Strength = 2MG
----
Product id = 6180
Application Number = 18226
Date of Application = 28,, Feb, 1983
RX/OTC/DISCN = DISCN
Tradename = BUMEX
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = VALIDUS PHARMS INC
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG/ML
----
Product id = 6177
Application Number = 74160
Date of Application = 30,, Oct, 1997
RX/OTC/DISCN = DISCN
Tradename = BUMETANIDE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG/ML
----
Product id = 18638
Application Number = 74225
Date of Application = 24,, Apr, 1995
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IVAX SUB TEVA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 0.5MG
----
Product id = 18639
Application Number = 74225
Date of Application = 24,, Apr, 1995
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IVAX SUB TEVA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 1MG
----
Product id = 18640
Application Number = 74225
Date of Application = 24,, Apr, 1995
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IVAX SUB TEVA PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 2MG
----
Product id = 6178
Application Number = 74332
Date of Application = 31,, Oct, 1994
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 0.25MG/ML
----
Product id = 6175
Application Number = 74441
Date of Application = 27,, Jan, 1995
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = BEDFORD
ProductNo = 001
Tecode = AP
Rld = Yes
Strength = 0.25MG/ML
----
Product id = 6179
Application Number = 74613
Date of Application = 18,, Nov, 1997
RX/OTC/DISCN = DISCN
Tradename = BUMETANIDE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = TEVA PARENTERAL
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG/ML
----
Product id = 18641
Application Number = 74700
Date of Application = 21,, Nov, 1996
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode = AB
Rld = No
Strength = 0.5MG
----
Product id = 18642
Application Number = 74700
Date of Application = 21,, Nov, 1996
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 002
Tecode = AB
Rld = No
Strength = 1MG
----
Product id = 18643
Application Number = 74700
Date of Application = 21,, Nov, 1996
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 003
Tecode = AB
Rld = Yes
Strength = 2MG
----
Product id = 6176
Application Number = 79196
Date of Application = 30,, Apr, 2008
RX/OTC/DISCN = RX
Tradename = BUMETANIDE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HIKMA MAPLE
ProductNo = 001
Tecode = AP
Rld = No
Strength = 0.25MG/ML
----