cetirizine-hydrochloride

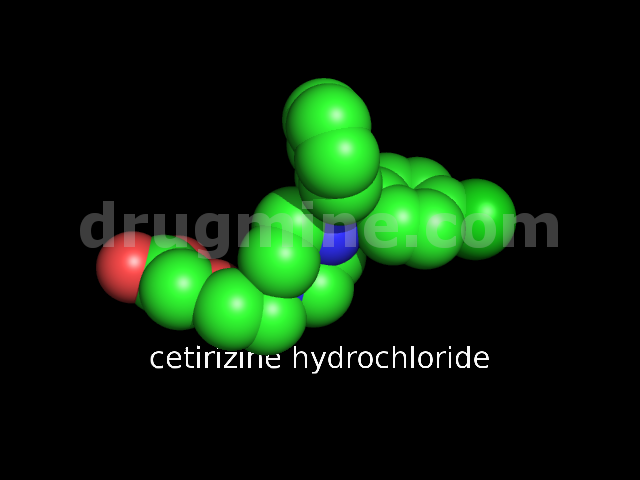
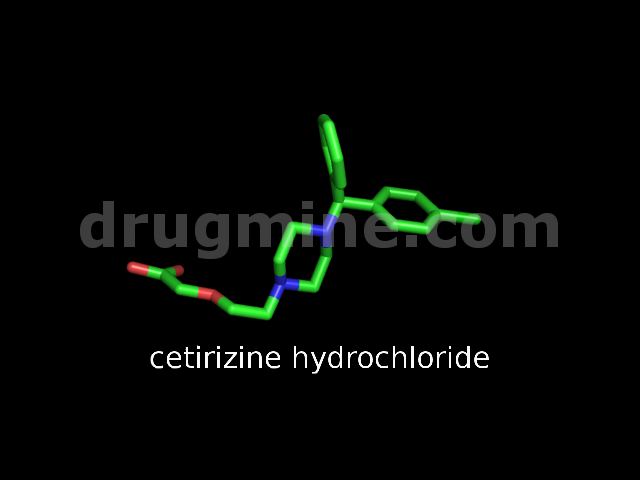
Name: CETIRIZINE HYDROCHLORIDE
ID :
MW: 389
Number of atoms: 27
Molecular_Formula: C21H25ClN2O3
Alogp: 0.453
Indication class : Antihistaminic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
cetirizine hydrochloride containing products summary
There are in total 139 different products containing the active ingredient cetirizine hydrochloride. From the 139 drug products, 20 have been discontinued.Product id = 29889
Application Number = 19835
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = ZYRTEC ALLERGY
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 29890
Application Number = 19835
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = ZYRTEC ALLERGY
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 004
Tecode =
Rld = Yes
Strength = 10MG
----
Product id = 29891
Application Number = 19835
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = ZYRTEC HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 005
Tecode =
Rld = No
Strength = 5MG
----
Product id = 29892
Application Number = 19835
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = ZYRTEC HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 006
Tecode =
Rld = Yes
Strength = 10MG
----
Product id = 15186
Application Number = 20346
Date of Application = 27,, Sep, 1996
RX/OTC/DISCN = RX
Tradename = ZYRTEC
Route/format = ORAL / SYRUP
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 001
Tecode = AA
Rld = Yes
Strength = 5MG/5ML
----
Product id = 16810
Application Number = 21150
Date of Application = 9,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = ZYRTEC-D 12 HOUR
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = N
Applicant Name = MCNEIL
ProductNo = 002
Tecode =
Rld = Yes
Strength = 5MG;120MG
----
Product id = 15243
Application Number = 21621
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S ZYRTEC ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = MCNEIL CONS
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 15244
Application Number = 21621
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S ZYRTEC ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = MCNEIL CONS
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 15245
Application Number = 21621
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S ZYRTEC HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = MCNEIL CONS
ProductNo = 005
Tecode =
Rld = No
Strength = 5MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 15246
Application Number = 21621
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S ZYRTEC HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = MCNEIL CONS
ProductNo = 006
Tecode =
Rld = No
Strength = 10MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 14958
Application Number = 22155
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S ZYRTEC HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 001
Tecode =
Rld = Yes
Strength = 5MG/5ML
----
Product id = 14957
Application Number = 22155
Date of Application = 16,, Nov, 2007
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S ZYRTEC ALLERGY
Route/format = ORAL / SYRUP
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 002
Tecode =
Rld = Yes
Strength = 5MG/5ML
----
Product id = 1325
Application Number = 22429
Date of Application = 23,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = BANNER LIFE SCIENCES
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 1328
Application Number = 22429
Date of Application = 23,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = BANNER LIFE SCIENCES
ProductNo = 002
Tecode =
Rld = Yes
Strength = 10MG
----
Product id = 1327
Application Number = 22429
Date of Application = 23,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = BANNER LIFE SCIENCES
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 1326
Application Number = 22429
Date of Application = 23,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = BANNER LIFE SCIENCES
ProductNo = 004
Tecode =
Rld = Yes
Strength = 10MG
----
Product id = 17125
Application Number = 22578
Date of Application = 3,, Sep, 2010
RX/OTC/DISCN = OTC
Tradename = ZYRTEC ALLERGY
Route/format = ORAL / TABLET, ORALLY DISINTEGRATING
Application Type = N
Applicant Name = MCNEIL CONSUMER
ProductNo = 001
Tecode =
Rld = Yes
Strength = 10MG
----
Product id = 19206
Application Number = 76047
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CONTRACT PHARMACAL
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19207
Application Number = 76047
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CONTRACT PHARMACAL
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14926
Application Number = 76601
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TARO
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 19212
Application Number = 76677
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19213
Application Number = 76677
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19241
Application Number = 76677
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 003
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19240
Application Number = 76677
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 004
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15805
Application Number = 77170
Date of Application = 25,, Feb, 2008
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE AND PSEUDOEPHEDRINE HYDROCHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = IVAX SUB TEVA PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG;120MG
----
Product id = 14927
Application Number = 77279
Date of Application = 27,, May, 2008
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 19204
Application Number = 77318
Date of Application = 25,, Jul, 2013
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19205
Application Number = 77318
Date of Application = 25,, Jul, 2013
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14923
Application Number = 77472
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = RANBAXY LABS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 19218
Application Number = 77498
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19219
Application Number = 77498
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19246
Application Number = 77498
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19247
Application Number = 77498
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = RANBAXY
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19222
Application Number = 77499
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19223
Application Number = 77499
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19248
Application Number = 77499
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19249
Application Number = 77499
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15228
Application Number = 77631
Date of Application = 11,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15229
Application Number = 77631
Date of Application = 11,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15227
Application Number = 77631
Date of Application = 11,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 003
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15226
Application Number = 77631
Date of Application = 11,, Jan, 2008
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 004
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19230
Application Number = 77829
Date of Application = 26,, Aug, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNIQUE PHARM LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19253
Application Number = 77829
Date of Application = 26,, Aug, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNIQUE PHARM LABS
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19252
Application Number = 77829
Date of Application = 26,, Aug, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNIQUE PHARM LABS
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19231
Application Number = 77829
Date of Application = 26,, Aug, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNIQUE PHARM LABS
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19220
Application Number = 77946
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19221
Application Number = 77946
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15806
Application Number = 77991
Date of Application = 5,, Mar, 2008
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE AND PSEUDOEPHEDRINE HYDROCHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG;120MG
----
Product id = 19224
Application Number = 78072
Date of Application = 22,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TARO
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19256
Application Number = 78072
Date of Application = 22,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TARO
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19225
Application Number = 78072
Date of Application = 22,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TARO
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19257
Application Number = 78072
Date of Application = 22,, Jul, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TARO
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19200
Application Number = 78317
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19201
Application Number = 78317
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19216
Application Number = 78336
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19217
Application Number = 78336
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19244
Application Number = 78336
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19245
Application Number = 78336
Date of Application = 27,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19236
Application Number = 78343
Date of Application = 15,, Jan, 2008
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19237
Application Number = 78343
Date of Application = 15,, Jan, 2008
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19209
Application Number = 78343
Date of Application = 15,, Jan, 2008
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 003
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19208
Application Number = 78343
Date of Application = 15,, Jan, 2008
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 004
Tecode =
Rld = No
Strength = 5MG
----
Product id = 14922
Application Number = 78398
Date of Application = 17,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 14918
Application Number = 78412
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 19232
Application Number = 78427
Date of Application = 28,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19233
Application Number = 78427
Date of Application = 28,, Dec, 2007
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14921
Application Number = 78488
Date of Application = 6,, Oct, 2008
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = BRECKENRIDGE PHARM
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 14928
Application Number = 78496
Date of Application = 25,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = VINTAGE
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 19196
Application Number = 78615
Date of Application = 28,, Dec, 2007
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ACTAVIS ELIZABETH
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19197
Application Number = 78615
Date of Application = 28,, Dec, 2007
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ACTAVIS ELIZABETH
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14916
Application Number = 78617
Date of Application = 2,, Feb, 2010
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = ACTAVIS MID ATLANTIC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 19250
Application Number = 78680
Date of Application = 26,, Jun, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19251
Application Number = 78680
Date of Application = 26,, Jun, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19228
Application Number = 78680
Date of Application = 26,, Jun, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19229
Application Number = 78680
Date of Application = 26,, Jun, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UNICHEM
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15233
Application Number = 78692
Date of Application = 14,, Feb, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15234
Application Number = 78692
Date of Application = 14,, Feb, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 002
Tecode =
Rld = Yes
Strength = 10MG
----
Product id = 14929
Application Number = 78757
Date of Application = 28,, Aug, 2009
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 19198
Application Number = 78780
Date of Application = 21,, Jan, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19255
Application Number = 78780
Date of Application = 21,, Jan, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19254
Application Number = 78780
Date of Application = 21,, Jan, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19199
Application Number = 78780
Date of Application = 21,, Jan, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AMNEAL PHARMS NY
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19214
Application Number = 78862
Date of Application = 19,, Feb, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19215
Application Number = 78862
Date of Application = 19,, Feb, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19242
Application Number = 78862
Date of Application = 19,, Feb, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19243
Application Number = 78862
Date of Application = 19,, Feb, 2009
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14920
Application Number = 78870
Date of Application = 27,, Apr, 2009
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = BIO PHARM INC
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 14924
Application Number = 78876
Date of Application = 11,, May, 2012
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SILARX
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 19202
Application Number = 78933
Date of Application = 15,, Jun, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CADISTA PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19203
Application Number = 78933
Date of Application = 15,, Jun, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CADISTA PHARMS
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19234
Application Number = 78933
Date of Application = 15,, Jun, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CADISTA PHARMS
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19235
Application Number = 78933
Date of Application = 15,, Jun, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CADISTA PHARMS
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19226
Application Number = 79191
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LLC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19259
Application Number = 79191
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LLC
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19258
Application Number = 79191
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LLC
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19227
Application Number = 79191
Date of Application = 15,, Apr, 2010
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TORRENT PHARMS LLC
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15235
Application Number = 90142
Date of Application = 30,, Aug, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARMA GLOBAL
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15236
Application Number = 90142
Date of Application = 30,, Aug, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARMA GLOBAL
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15239
Application Number = 90142
Date of Application = 30,, Aug, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARMA GLOBAL
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15240
Application Number = 90142
Date of Application = 30,, Aug, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = SUN PHARMA GLOBAL
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14954
Application Number = 90182
Date of Application = 22,, Apr, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TARO
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14941
Application Number = 90182
Date of Application = 22,, Apr, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TARO
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14951
Application Number = 90183
Date of Application = 24,, Apr, 2008
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = RANBAXY LABS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14938
Application Number = 90183
Date of Application = 24,, Apr, 2008
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = RANBAXY LABS LTD
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14946
Application Number = 90188
Date of Application = 22,, Apr, 2008
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14932
Application Number = 90188
Date of Application = 22,, Apr, 2008
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14925
Application Number = 90191
Date of Application = 12,, Nov, 2009
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 14950
Application Number = 90254
Date of Application = 9,, Apr, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14937
Application Number = 90254
Date of Application = 9,, Apr, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14935
Application Number = 90300
Date of Application = 10,, Oct, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = CYPRESS PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14949
Application Number = 90300
Date of Application = 10,, Oct, 2008
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = CYPRESS PHARM
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14944
Application Number = 90378
Date of Application = 9,, May, 2008
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = ACTAVIS MID ATLANTIC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14930
Application Number = 90378
Date of Application = 9,, May, 2008
RX/OTC/DISCN = DISCN
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = ACTAVIS MID ATLANTIC
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14948
Application Number = 90474
Date of Application = 30,, Mar, 2009
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = BIO PHARM INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14934
Application Number = 90474
Date of Application = 30,, Mar, 2009
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = BIO PHARM INC
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14943
Application Number = 90572
Date of Application = 16,, Nov, 2012
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TRIS PHARMA INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14956
Application Number = 90572
Date of Application = 16,, Nov, 2012
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TRIS PHARMA INC
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14947
Application Number = 90750
Date of Application = 2,, Feb, 2010
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14933
Application Number = 90750
Date of Application = 2,, Feb, 2010
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14919
Application Number = 90751
Date of Application = 16,, Dec, 2009
RX/OTC/DISCN = DISCN
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14945
Application Number = 90765
Date of Application = 7,, Oct, 2009
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = AMNEAL PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14931
Application Number = 90765
Date of Application = 7,, Oct, 2009
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = AMNEAL PHARMS
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14917
Application Number = 90766
Date of Application = 7,, Oct, 2009
RX/OTC/DISCN = RX
Tradename = CETIRIZINE HYDROCHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = AMNEAL PHARMS
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 15807
Application Number = 90922
Date of Application = 28,, Sep, 2012
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE AND PSEUDOEPHEDRINE HYDROCHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = SUN PHARM INDS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG;120MG
----
Product id = 15231
Application Number = 91116
Date of Application = 19,, Feb, 2015
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = JUBILANT GENERICS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15232
Application Number = 91116
Date of Application = 19,, Feb, 2015
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = JUBILANT GENERICS
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 15237
Application Number = 91116
Date of Application = 19,, Feb, 2015
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = JUBILANT GENERICS
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15238
Application Number = 91116
Date of Application = 19,, Feb, 2015
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / TABLET, CHEWABLE
Application Type = A
Applicant Name = JUBILANT GENERICS
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14939
Application Number = 91130
Date of Application = 22,, Apr, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SILARX
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14952
Application Number = 91130
Date of Application = 22,, Apr, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SILARX
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14940
Application Number = 91327
Date of Application = 17,, Oct, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14953
Application Number = 91327
Date of Application = 17,, Oct, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SUN PHARM INDS INC
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14942
Application Number = 201546
Date of Application = 20,, May, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TARO
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 14955
Application Number = 201546
Date of Application = 20,, May, 2011
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE HIVES RELIEF
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = TARO
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 19238
Application Number = 202277
Date of Application = 11,, Mar, 2014
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IPCA LABS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19210
Application Number = 202277
Date of Application = 11,, Mar, 2014
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IPCA LABS LTD
ProductNo = 002
Tecode =
Rld = No
Strength = 5MG
----
Product id = 19239
Application Number = 202277
Date of Application = 11,, Mar, 2014
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE HIVES
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IPCA LABS LTD
ProductNo = 003
Tecode =
Rld = No
Strength = 10MG
----
Product id = 19211
Application Number = 202277
Date of Application = 11,, Mar, 2014
RX/OTC/DISCN = OTC
Tradename = CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IPCA LABS LTD
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 14936
Application Number = 204226
Date of Application = 9,, Sep, 2013
RX/OTC/DISCN = OTC
Tradename = CHILDREN'S CETIRIZINE HYDROCHLORIDE ALLERGY
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = PERRIGO R AND D
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----