dexamethasone
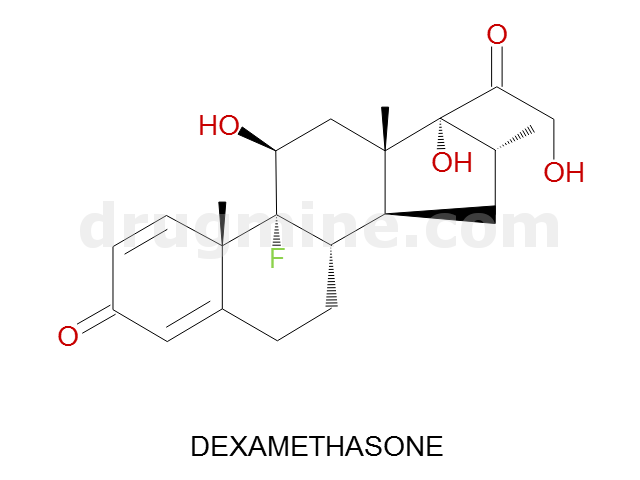

Name: DEXAMETHASONE
ID :
MW: 392
Number of atoms: 28
Molecular_Formula: C22H29FO5
Alogp: 1.708
Indication class : Glucocorticoid
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
dexamethasone containing products summary
There are in total 82 different products containing the active ingredient dexamethasone. From the 82 drug products, 50 have been discontinued.Product id = 20132
Application Number = 11664
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MERCK
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 20133
Application Number = 11664
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MERCK
ProductNo = 002
Tecode =
Rld = No
Strength = 0.75MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 20134
Application Number = 11664
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MERCK
ProductNo = 003
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20131
Application Number = 11664
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MERCK
ProductNo = 004
Tecode =
Rld = No
Strength = 0.25MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 20135
Application Number = 11664
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MERCK
ProductNo = 005
Tecode =
Rld = No
Strength = 4MG
----
Product id = 20136
Application Number = 11664
Date of Application = 30,, Jul, 1982
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = MERCK
ProductNo = 006
Tecode =
Rld = No
Strength = 6MG
----
Product id = 4529
Application Number = 12376
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADRON
Route/format = ORAL / ELIXIR
Application Type = N
Applicant Name = MERCK
ProductNo = 002
Tecode =
Rld = No
Strength = 0.5MG/5ML
----
Product id = 4554
Application Number = 12674
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXADROL
Route/format = ORAL / ELIXIR
Application Type = N
Applicant Name = ORGANON USA INC
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG/5ML
----
Product id = 22151
Application Number = 12675
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXADROL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = ORGANON USA INC
ProductNo = 004
Tecode =
Rld = No
Strength = 0.5MG
----
Product id = 22152
Application Number = 12675
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXADROL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = ORGANON USA INC
ProductNo = 007
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 22153
Application Number = 12675
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXADROL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = ORGANON USA INC
ProductNo = 009
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 22154
Application Number = 12675
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXADROL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = ORGANON USA INC
ProductNo = 010
Tecode =
Rld = No
Strength = 4MG
----
Product id = 101
Application Number = 12731
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECASPRAY
Route/format = TOPICAL / AEROSOL
Application Type = N
Applicant Name = MERCK
ProductNo = 002
Tecode =
Rld = No
Strength = 0.04% **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 14560
Application Number = 13422
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = MAXIDEX
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = ALCON
ProductNo = 001
Tecode =
Rld = Yes
Strength = 0.1%
----
Product id = 5386
Application Number = 13538
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DECADERM
Route/format = TOPICAL / GEL
Application Type = N
Applicant Name = MERCK
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%
----
Product id = 14595
Application Number = 21537
Date of Application = 18,, Jul, 2003
RX/OTC/DISCN = RX
Tradename = CIPRODEX
Route/format = OTIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = ALCON PHARMS LTD
ProductNo = 001
Tecode =
Rld = Yes
Strength = 0.3%;0.1%
----
Product id = 5546
Application Number = 22315
Date of Application = 17,, Jun, 2009
RX/OTC/DISCN = RX
Tradename = OZURDEX
Route/format = INTRAVITREAL / IMPLANT
Application Type = N
Applicant Name = ALLERGAN
ProductNo = 001
Tecode =
Rld = Yes
Strength = 0.7MG
----
Product id = 20216
Application Number = 40700
Date of Application = 15,, Aug, 2008
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ECR
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.5MG
----
Product id = 14562
Application Number = 50023
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = MAXITROL
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = FALCON PHARMS
ProductNo = 002
Tecode = AT
Rld = Yes
Strength = 0.1%;EQ 3.5MG BASE/ML;10,000 UNITS/ML
----
Product id = 12345
Application Number = 50065
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = MAXITROL
Route/format = OPHTHALMIC / OINTMENT
Application Type = N
Applicant Name = FALCON PHARMS
ProductNo = 002
Tecode = AT
Rld = Yes
Strength = 0.1%;EQ 3.5MG BASE/GM;10,000 UNITS/GM
----
Product id = 14581
Application Number = 50592
Date of Application = 18,, Aug, 1988
RX/OTC/DISCN = RX
Tradename = TOBRADEX
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = ALCON
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = 0.1%;0.3%
----
Product id = 12375
Application Number = 50616
Date of Application = 28,, Sep, 1988
RX/OTC/DISCN = RX
Tradename = TOBRADEX
Route/format = OPHTHALMIC / OINTMENT
Application Type = N
Applicant Name = ALCON
ProductNo = 001
Tecode =
Rld = Yes
Strength = 0.1%;0.3%
----
Product id = 14582
Application Number = 50818
Date of Application = 13,, Feb, 2009
RX/OTC/DISCN = RX
Tradename = TOBRADEX ST
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = N
Applicant Name = ALCON PHARMS LTD
ProductNo = 001
Tecode =
Rld = Yes
Strength = 0.05%;0.3%
----
Product id = 14561
Application Number = 62341
Date of Application = 22,, May, 1984
RX/OTC/DISCN = RX
Tradename = MAXITROL
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = ALCON
ProductNo = 001
Tecode = AT
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/ML;10,000 UNITS/ML
----
Product id = 12326
Application Number = 62411
Date of Application = 16,, May, 1983
RX/OTC/DISCN = DISCN
Tradename = DEXASPORIN
Route/format = OPHTHALMIC / OINTMENT
Application Type = A
Applicant Name = PHARMAFAIR
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/GM;10,000 UNITS/GM
----
Product id = 14549
Application Number = 62428
Date of Application = 18,, May, 1983
RX/OTC/DISCN = DISCN
Tradename = DEXASPORIN
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = PHARMAFAIR
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/ML;10,000 UNITS/ML
----
Product id = 14546
Application Number = 62544
Date of Application = 29,, Oct, 1984
RX/OTC/DISCN = DISCN
Tradename = DEXACIDIN
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = NOVARTIS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/ML;10,000 UNITS/ML
----
Product id = 12324
Application Number = 62566
Date of Application = 22,, Feb, 1985
RX/OTC/DISCN = DISCN
Tradename = DEXACIDIN
Route/format = OPHTHALMIC / OINTMENT
Application Type = A
Applicant Name = NOVARTIS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/GM;10,000 UNITS/GM
----
Product id = 14567
Application Number = 62721
Date of Application = 17,, Nov, 1986
RX/OTC/DISCN = DISCN
Tradename = NEOMYCIN AND POLYMYXIN B SULFATES AND DEXAMETHASONE
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = ALCON PHARMS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/ML;10,000 UNITS/ML
----
Product id = 12360
Application Number = 62938
Date of Application = 31,, Jul, 1989
RX/OTC/DISCN = RX
Tradename = NEOMYCIN AND POLYMYXIN B SULFATES AND DEXAMETHASONE
Route/format = OPHTHALMIC / OINTMENT
Application Type = A
Applicant Name = PERRIGO CO TENNESSEE
ProductNo = 001
Tecode = AT
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/GM;10,000 UNITS/GM
----
Product id = 12359
Application Number = 64063
Date of Application = 25,, Jul, 1994
RX/OTC/DISCN = RX
Tradename = NEOMYCIN AND POLYMYXIN B SULFATES AND DEXAMETHASONE
Route/format = OPHTHALMIC / OINTMENT
Application Type = A
Applicant Name = BAUSCH AND LOMB
ProductNo = 001
Tecode = AT
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/GM;10,000 UNITS/GM
----
Product id = 14583
Application Number = 64134
Date of Application = 27,, Oct, 1999
RX/OTC/DISCN = RX
Tradename = TOBRAMYCIN AND DEXAMETHASONE
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = BAUSCH AND LOMB
ProductNo = 001
Tecode = AB
Rld = No
Strength = 0.1%;0.3%
----
Product id = 14548
Application Number = 64135
Date of Application = 13,, Sep, 1995
RX/OTC/DISCN = RX
Tradename = DEXASPORIN
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = BAUSCH AND LOMB
ProductNo = 001
Tecode = AT
Rld = No
Strength = 0.1%;EQ 3.5MG BASE/ML;10,000 UNITS/ML
----
Product id = 20242
Application Number = 80399
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20247
Application Number = 80968
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 99
Application Number = 83296
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = AEROSEB-DEX
Route/format = TOPICAL / AEROSOL
Application Type = A
Applicant Name = ALLERGAN HERBERT
ProductNo = 002
Tecode =
Rld = No
Strength = 0.01% **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 20233
Application Number = 83420
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PVT FORM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20232
Application Number = 83806
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PHOENIX LABS NY
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20218
Application Number = 84013
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG
----
Product id = 20222
Application Number = 84081
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20220
Application Number = 84084
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG
----
Product id = 20224
Application Number = 84086
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20252
Application Number = 84327
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WHITEWORTH TOWN PLSN
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20248
Application Number = 84457
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20238
Application Number = 84610
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.5MG
----
Product id = 20235
Application Number = 84611
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = BP
Rld = No
Strength = 0.5MG
----
Product id = 20240
Application Number = 84612
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = BP
Rld = No
Strength = 4MG
----
Product id = 20236
Application Number = 84613
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = BP
Rld = No
Strength = 0.75MG
----
Product id = 20234
Application Number = 84614
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG
----
Product id = 4532
Application Number = 84754
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / ELIXIR
Application Type = A
Applicant Name = STI PHARMA LLC
ProductNo = 001
Tecode = AA
Rld = Yes
Strength = 0.5MG/5ML
----
Product id = 20225
Application Number = 84763
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20219
Application Number = 84764
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG
----
Product id = 20223
Application Number = 84765
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20221
Application Number = 84766
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MUTUAL PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG
----
Product id = 20262
Application Number = 84990
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXONE 1.5
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SOLVAY
ProductNo = 001
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20260
Application Number = 84991
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXONE 0.5
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SOLVAY
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG
----
Product id = 20263
Application Number = 84992
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXONE 4
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SOLVAY
ProductNo = 001
Tecode =
Rld = No
Strength = 4MG
----
Product id = 20261
Application Number = 84993
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXONE 0.75
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SOLVAY
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20217
Application Number = 85376
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IMPAX LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20245
Application Number = 85455
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG
----
Product id = 20250
Application Number = 85456
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20246
Application Number = 85458
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG
----
Product id = 20249
Application Number = 85818
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20251
Application Number = 85840
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20244
Application Number = 87533
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 001
Tecode =
Rld = No
Strength = 1.5MG
----
Product id = 20243
Application Number = 87534
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 001
Tecode =
Rld = No
Strength = 0.75MG
----
Product id = 20239
Application Number = 87916
Date of Application = 26,, Aug, 1982
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = BP
Rld = No
Strength = 2MG
----
Product id = 20227
Application Number = 88148
Date of Application = 28,, Apr, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode = BP
Rld = No
Strength = 0.5MG
----
Product id = 20226
Application Number = 88149
Date of Application = 28,, Apr, 1983
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25MG
----
Product id = 20228
Application Number = 88160
Date of Application = 28,, Apr, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode = BP
Rld = No
Strength = 0.75MG
----
Product id = 20229
Application Number = 88237
Date of Application = 28,, Apr, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode = BP
Rld = No
Strength = 1.5MG
----
Product id = 20230
Application Number = 88238
Date of Application = 28,, Apr, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode = BP
Rld = No
Strength = 4MG
----
Product id = 13790
Application Number = 88248
Date of Application = 1,, Sep, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / SOLUTION
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode =
Rld = Yes
Strength = 0.5MG/5ML
----
Product id = 3919
Application Number = 88252
Date of Application = 1,, Sep, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE INTENSOL
Route/format = ORAL / CONCENTRATE
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode =
Rld = Yes
Strength = 1MG/ML
----
Product id = 4534
Application Number = 88254
Date of Application = 27,, Jul, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / ELIXIR
Application Type = A
Applicant Name = WOCKHARDT EU OPERATN
ProductNo = 001
Tecode = AA
Rld = No
Strength = 0.5MG/5ML
----
Product id = 20237
Application Number = 88306
Date of Application = 15,, Sep, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = BP
Rld = No
Strength = 1MG
----
Product id = 20241
Application Number = 88316
Date of Application = 15,, Sep, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = BP
Rld = Yes
Strength = 6MG
----
Product id = 20231
Application Number = 88481
Date of Application = 28,, Nov, 1983
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PAR PHARM
ProductNo = 001
Tecode = BP
Rld = Yes
Strength = 6MG
----
Product id = 4530
Application Number = 88997
Date of Application = 10,, Oct, 1986
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = ORAL / ELIXIR
Application Type = A
Applicant Name = ALPHARMA US PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.5MG/5ML
----
Product id = 14547
Application Number = 89170
Date of Application = 9,, May, 1989
RX/OTC/DISCN = DISCN
Tradename = DEXAMETHASONE
Route/format = OPHTHALMIC / SUSPENSION/DROPS
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.1%
----
Product id = 4531
Application Number = 90891
Date of Application = 12,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / ELIXIR
Application Type = A
Applicant Name = LYNE
ProductNo = 001
Tecode = AA
Rld = No
Strength = 0.5MG/5ML
----
Product id = 4533
Application Number = 91188
Date of Application = 11,, May, 2011
RX/OTC/DISCN = RX
Tradename = DEXAMETHASONE
Route/format = ORAL / ELIXIR
Application Type = A
Applicant Name = VINTAGE PHARMS
ProductNo = 001
Tecode = AA
Rld = No
Strength = 0.5MG/5ML
----