didanosine
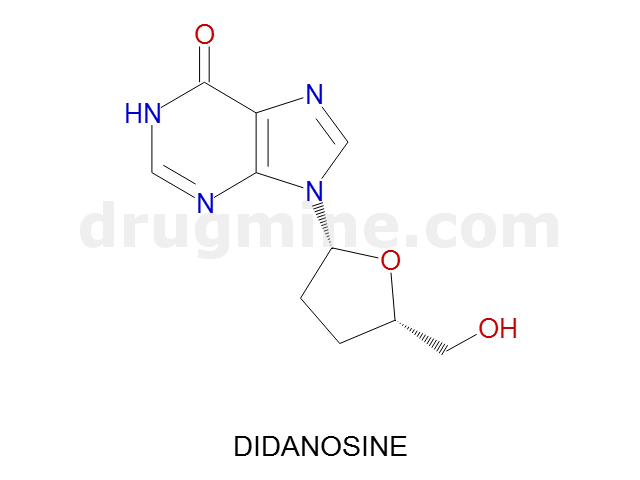


Name: DIDANOSINE
ID :
MW: 236
Number of atoms: 17
Molecular_Formula: C10H12N4O3
Alogp: -0.844
Indication class : Antiviral
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
didanosine containing products summary
There are in total 29 different products containing the active ingredient didanosine. From the 29 drug products, 9 have been discontinued.Product id = 15372
Application Number = 20154
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 002
Tecode =
Rld = No
Strength = 25MG
----
Product id = 15373
Application Number = 20154
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 003
Tecode =
Rld = No
Strength = 50MG
----
Product id = 15374
Application Number = 20154
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 004
Tecode =
Rld = No
Strength = 100MG
----
Product id = 15375
Application Number = 20154
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 005
Tecode =
Rld = No
Strength = 150MG
----
Product id = 15376
Application Number = 20154
Date of Application = 28,, Oct, 1999
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / TABLET, CHEWABLE
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 006
Tecode =
Rld = No
Strength = 200MG
----
Product id = 4952
Application Number = 20155
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / FOR SOLUTION
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 003
Tecode =
Rld = No
Strength = 100MG/PACKET
----
Product id = 4953
Application Number = 20155
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / FOR SOLUTION
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 004
Tecode =
Rld = No
Strength = 167MG/PACKET
----
Product id = 4954
Application Number = 20155
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / FOR SOLUTION
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 005
Tecode =
Rld = No
Strength = 250MG/PACKET
----
Product id = 4955
Application Number = 20155
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = DISCN
Tradename = VIDEX
Route/format = ORAL / FOR SOLUTION
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 006
Tecode =
Rld = No
Strength = 375MG/PACKET
----
Product id = 4951
Application Number = 20156
Date of Application = 9,, Oct, 1991
RX/OTC/DISCN = RX
Tradename = VIDEX
Route/format = ORAL / FOR SOLUTION
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 001
Tecode = AA
Rld = Yes
Strength = 10MG/ML
----
Product id = 243
Application Number = 21183
Date of Application = 31,, Oct, 2000
RX/OTC/DISCN = RX
Tradename = VIDEX EC
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 244
Application Number = 21183
Date of Application = 31,, Oct, 2000
RX/OTC/DISCN = RX
Tradename = VIDEX EC
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 245
Application Number = 21183
Date of Application = 31,, Oct, 2000
RX/OTC/DISCN = RX
Tradename = VIDEX EC
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 003
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 246
Application Number = 21183
Date of Application = 31,, Oct, 2000
RX/OTC/DISCN = RX
Tradename = VIDEX EC
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = N
Applicant Name = BRISTOL MYERS SQUIBB
ProductNo = 004
Tecode = AB
Rld = Yes
Strength = 400MG
----
Product id = 124
Application Number = 77167
Date of Application = 3,, Dec, 2004
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = BARR
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 125
Application Number = 77167
Date of Application = 3,, Dec, 2004
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = BARR
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 126
Application Number = 77167
Date of Application = 3,, Dec, 2004
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = BARR
ProductNo = 003
Tecode = AB
Rld = No
Strength = 400MG
----
Product id = 16819
Application Number = 77275
Date of Application = 14,, Aug, 2012
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / TABLET, FOR SUSPENSION
Application Type = A
Applicant Name = AUROBINDO
ProductNo = 001
Tecode =
Rld = No
Strength = 100MG
----
Product id = 16820
Application Number = 77275
Date of Application = 14,, Aug, 2012
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / TABLET, FOR SUSPENSION
Application Type = A
Applicant Name = AUROBINDO
ProductNo = 002
Tecode =
Rld = No
Strength = 150MG
----
Product id = 16821
Application Number = 77275
Date of Application = 14,, Aug, 2012
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / TABLET, FOR SUSPENSION
Application Type = A
Applicant Name = AUROBINDO
ProductNo = 003
Tecode =
Rld = Yes
Strength = 200MG
----
Product id = 4855
Application Number = 78112
Date of Application = 8,, Mar, 2007
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / FOR SOLUTION
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 001
Tecode = AA
Rld = No
Strength = 10MG/ML
----
Product id = 120
Application Number = 90094
Date of Application = 24,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 121
Application Number = 90094
Date of Application = 24,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 122
Application Number = 90094
Date of Application = 24,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 003
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 123
Application Number = 90094
Date of Application = 24,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 004
Tecode = AB
Rld = No
Strength = 400MG
----
Product id = 127
Application Number = 90788
Date of Application = 8,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 128
Application Number = 90788
Date of Application = 8,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 129
Application Number = 90788
Date of Application = 8,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 130
Application Number = 90788
Date of Application = 8,, Apr, 2010
RX/OTC/DISCN = RX
Tradename = DIDANOSINE
Route/format = ORAL / CAPSULE, DELAYED REL PELLETS
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = 400MG
----