dolasetron-mesylate

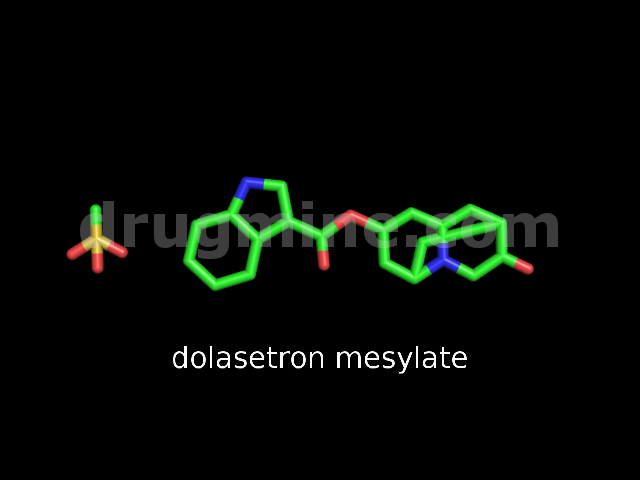
Name: DOLASETRON MESYLATE
ID :
MW: 324
Number of atoms: 24
Molecular_Formula: C19H20N2O3
Alogp: 2.035
Indication class : Antimigraine; Anti-Emetic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
dolasetron mesylate containing products summary
There are in total 5 different products containing the active ingredient dolasetron mesylate. From the 5 drug products, zero have been discontinued.Product id = 18045
Application Number = 20623
Date of Application = 11,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = ANZEMET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = No
Strength = 50MG
----
Product id = 18046
Application Number = 20623
Date of Application = 11,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = ANZEMET
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 002
Tecode =
Rld = Yes
Strength = 100MG
----
Product id = 5989
Application Number = 20624
Date of Application = 11,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = ANZEMET
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = Yes
Strength = 100MG/5ML (20MG/ML)
----
Product id = 5988
Application Number = 20624
Date of Application = 11,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = ANZEMET
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 002
Tecode =
Rld = Yes
Strength = 12.5MG/0.625ML (20MG/ML)
----
Product id = 5990
Application Number = 20624
Date of Application = 11,, Dec, 2001
RX/OTC/DISCN = RX
Tradename = ANZEMET
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 003
Tecode =
Rld = Yes
Strength = 500MG/25ML (20MG/ML)
----