droperidol
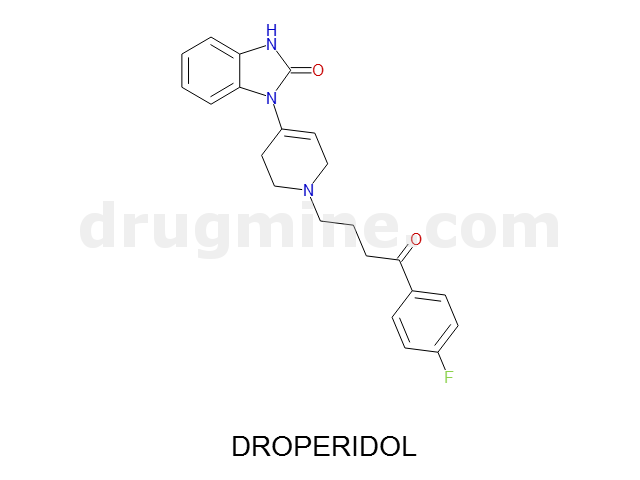

Name: DROPERIDOL
ID :
MW: 379
Number of atoms: 28
Molecular_Formula: C22H22FN3O2
Alogp: 3.049
Indication class : Antipsychotic
Oral Flag : 0
Max_Phase : 4
Molecule_Type : Small molecule
droperidol containing products summary
There are in total 23 different products containing the active ingredient droperidol. From the 23 drug products, 20 have been discontinued.Product id = 8411
Application Number = 16049
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = INNOVAR
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = AKORN MFG
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML;EQ 0.05MG BASE/ML
----
Product id = 8397
Application Number = 16796
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = INAPSINE
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = AKORN INC
ProductNo = 001
Tecode = AP
Rld = Yes
Strength = 2.5MG/ML
----
Product id = 7423
Application Number = 70992
Date of Application = 17,, Nov, 1986
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ABRAXIS PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7424
Application Number = 70993
Date of Application = 17,, Nov, 1986
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ABRAXIS PHARM
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7429
Application Number = 71645
Date of Application = 7,, Apr, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7434
Application Number = 71750
Date of Application = 6,, Sep, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = SMITH AND NEPHEW
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7435
Application Number = 71754
Date of Application = 6,, Sep, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = SOLOPAK
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7436
Application Number = 71755
Date of Application = 6,, Sep, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = SOLOPAK
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7430
Application Number = 71981
Date of Application = 29,, Feb, 1988
RX/OTC/DISCN = RX
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 2.5MG/ML
----
Product id = 7610
Application Number = 71982
Date of Application = 4,, May, 1988
RX/OTC/DISCN = DISCN
Tradename = FENTANYL CITRATE AND DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML;EQ 0.05MG BASE/ML
----
Product id = 7425
Application Number = 72018
Date of Application = 20,, Oct, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7426
Application Number = 72019
Date of Application = 19,, Oct, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7427
Application Number = 72020
Date of Application = 19,, Oct, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7428
Application Number = 72021
Date of Application = 19,, Oct, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7607
Application Number = 72026
Date of Application = 13,, Apr, 1989
RX/OTC/DISCN = DISCN
Tradename = FENTANYL CITRATE AND DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML;EQ 0.05MG BASE/ML
----
Product id = 7608
Application Number = 72027
Date of Application = 13,, Apr, 1989
RX/OTC/DISCN = DISCN
Tradename = FENTANYL CITRATE AND DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML;EQ 0.05MG BASE/ML
----
Product id = 7609
Application Number = 72028
Date of Application = 13,, Apr, 1989
RX/OTC/DISCN = DISCN
Tradename = FENTANYL CITRATE AND DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ASTRAZENECA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML;EQ 0.05MG BASE/ML
----
Product id = 7432
Application Number = 72123
Date of Application = 24,, Oct, 1988
RX/OTC/DISCN = RX
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = LUITPOLD
ProductNo = 001
Tecode = AP
Rld = No
Strength = 2.5MG/ML
----
Product id = 7431
Application Number = 72272
Date of Application = 31,, Aug, 1995
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7433
Application Number = 72335
Date of Application = 24,, Oct, 1988
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = LUITPOLD
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7437
Application Number = 73520
Date of Application = 27,, Nov, 1991
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7438
Application Number = 73521
Date of Application = 27,, Nov, 1991
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----
Product id = 7439
Application Number = 73523
Date of Application = 27,, Nov, 1991
RX/OTC/DISCN = DISCN
Tradename = DROPERIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 2.5MG/ML
----