famciclovir
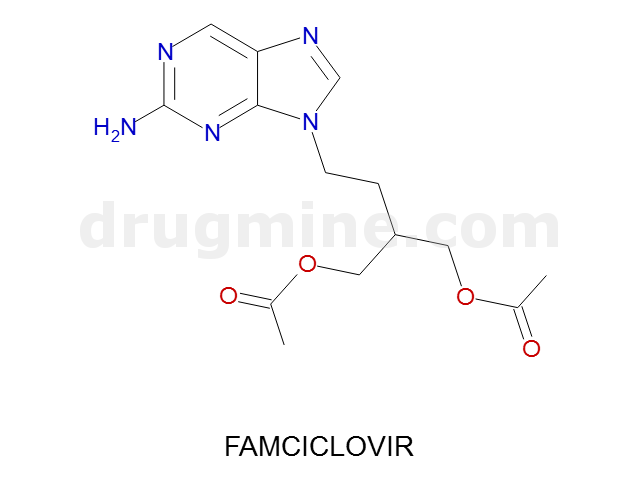
Name: FAMCICLOVIR
ID :
MW: 321
Number of atoms: 23
Molecular_Formula: C14H19N5O4
Alogp: -0.294
Indication class : Antiviral
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
famciclovir containing products summary
There are in total 27 different products containing the active ingredient famciclovir. From the 27 drug products, zero have been discontinued.Product id = 21211
Application Number = 20363
Date of Application = 26,, Apr, 1996
RX/OTC/DISCN = RX
Tradename = FAMVIR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21212
Application Number = 20363
Date of Application = 29,, Jun, 1994
RX/OTC/DISCN = RX
Tradename = FAMVIR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode = AB
Rld = Yes
Strength = 500MG
----
Product id = 21210
Application Number = 20363
Date of Application = 11,, Dec, 1995
RX/OTC/DISCN = RX
Tradename = FAMVIR
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21161
Application Number = 77487
Date of Application = 24,, Aug, 2007
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21162
Application Number = 77487
Date of Application = 24,, Aug, 2007
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21163
Application Number = 77487
Date of Application = 24,, Aug, 2007
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21146
Application Number = 78278
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21147
Application Number = 78278
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21148
Application Number = 78278
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21158
Application Number = 90128
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21159
Application Number = 90128
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21160
Application Number = 90128
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ROXANE
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21143
Application Number = 91114
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21144
Application Number = 91114
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21145
Application Number = 91114
Date of Application = 21,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21140
Application Number = 91480
Date of Application = 22,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21141
Application Number = 91480
Date of Application = 22,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21142
Application Number = 91480
Date of Application = 22,, Jul, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21152
Application Number = 201022
Date of Application = 12,, Jan, 2012
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MACLEODS PHARMS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21153
Application Number = 201022
Date of Application = 12,, Jan, 2012
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MACLEODS PHARMS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21154
Application Number = 201022
Date of Application = 12,, Jan, 2012
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MACLEODS PHARMS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21155
Application Number = 201333
Date of Application = 24,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21156
Application Number = 201333
Date of Application = 24,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21157
Application Number = 201333
Date of Application = 24,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----
Product id = 21149
Application Number = 202438
Date of Application = 10,, Sep, 2014
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HETERO LABS LTD V
ProductNo = 001
Tecode = AB
Rld = No
Strength = 125MG
----
Product id = 21150
Application Number = 202438
Date of Application = 10,, Sep, 2014
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HETERO LABS LTD V
ProductNo = 002
Tecode = AB
Rld = No
Strength = 250MG
----
Product id = 21151
Application Number = 202438
Date of Application = 10,, Sep, 2014
RX/OTC/DISCN = RX
Tradename = FAMCICLOVIR
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HETERO LABS LTD V
ProductNo = 003
Tecode = AB
Rld = No
Strength = 500MG
----