fludarabine-phosphate
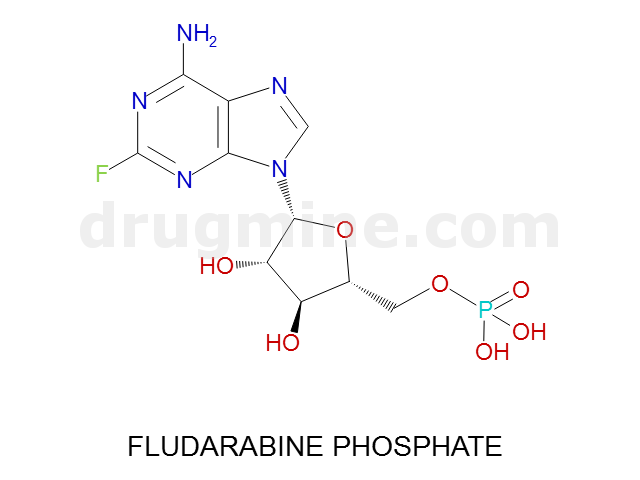


Name: FLUDARABINE PHOSPHATE
ID :
MW: 365
Number of atoms: 24
Molecular_Formula: C10H13FN5O7P
Alogp: -1.384
Indication class : Antineoplastic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
fludarabine phosphate containing products summary
There are in total 12 different products containing the active ingredient fludarabine phosphate. From the 12 drug products, 2 have been discontinued.Product id = 7667
Application Number = 20038
Date of Application = 18,, Apr, 1991
RX/OTC/DISCN = DISCN
Tradename = FLUDARA
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = GENZYME CORP
ProductNo = 001
Tecode =
Rld = No
Strength = 50MG/VIAL
----
Product id = 7675
Application Number = 22137
Date of Application = 21,, Sep, 2007
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = SANDOZ
ProductNo = 001
Tecode = AP
Rld = Yes
Strength = 50MG/2ML (25MG/ML)
----
Product id = 25543
Application Number = 22273
Date of Application = 18,, Dec, 2008
RX/OTC/DISCN = DISCN
Tradename = OFORTA
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = No
Strength = 10MG
----
Product id = 7676
Application Number = 76349
Date of Application = 28,, Aug, 2003
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = TEVA PHARMS USA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/VIAL
----
Product id = 7677
Application Number = 76661
Date of Application = 28,, Apr, 2004
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = TEVA PHARMS USA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/2ML (25MG/ML)
----
Product id = 7671
Application Number = 77790
Date of Application = 6,, Apr, 2007
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode = AP
Rld = Yes
Strength = 50MG/VIAL
----
Product id = 7669
Application Number = 78393
Date of Application = 15,, Oct, 2007
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = FRESENIUS KABI USA
ProductNo = 001
Tecode = AP
Rld = Yes
Strength = 50MG/2ML (25MG/ML)
----
Product id = 7670
Application Number = 78544
Date of Application = 15,, Oct, 2007
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = FRESENIUS KABI USA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/VIAL
----
Product id = 7668
Application Number = 78610
Date of Application = 11,, Feb, 2009
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ACTAVIS TOTOWA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/VIAL
----
Product id = 7672
Application Number = 90724
Date of Application = 27,, Sep, 2010
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = MUSTAFA NEVZAT ILAC
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/2ML (25MG/ML)
----
Product id = 7673
Application Number = 200647
Date of Application = 21,, Dec, 2011
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ONCO THERAPIES LTD
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/2ML (25MG/ML)
----
Product id = 7674
Application Number = 200648
Date of Application = 16,, Oct, 2012
RX/OTC/DISCN = RX
Tradename = FLUDARABINE PHOSPHATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ONCO THERAPIES LTD
ProductNo = 001
Tecode = AP
Rld = No
Strength = 50MG/VIAL
----