hexachlorophene

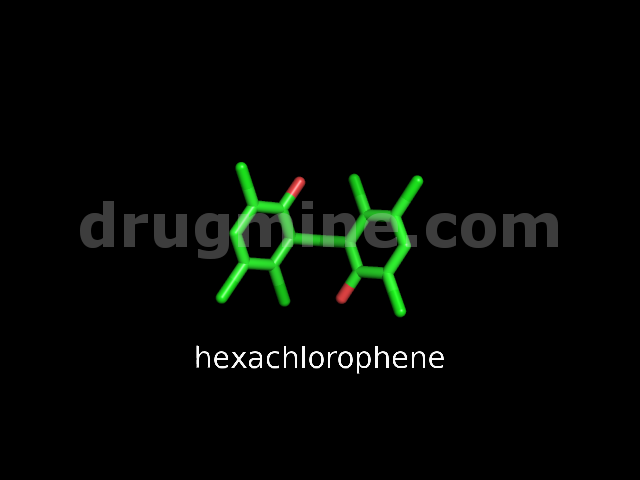
Name: HEXACHLOROPHENE
ID :
MW: 407
Number of atoms: 21
Molecular_Formula: C13H6Cl6O2
Alogp: 7.307
Indication class : Anti-Infective, Topical; Detergent
Oral Flag : 0
Max_Phase : 4
Molecule_Type : Small molecule
hexachlorophene containing products summary
There are in total 19 different products containing the active ingredient hexachlorophene. From the 19 drug products, 16 have been discontinued.Product id = 12829
Application Number = 6270
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = GAMOPHEN
Route/format = TOPICAL / SOAP
Application Type = N
Applicant Name = ARBROOK
ProductNo = 003
Tecode =
Rld = No
Strength = 2%
----
Product id = 4596
Application Number = 6882
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = PHISOHEX
Route/format = TOPICAL / EMULSION
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = Yes
Strength = 3%
----
Product id = 4597
Application Number = 8402
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = PHISOHEX
Route/format = TOPICAL / EMULSION
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----
Product id = 4598
Application Number = 17405
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = SOY-DOME
Route/format = TOPICAL / EMULSION
Application Type = N
Applicant Name = BAYER PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----
Product id = 4595
Application Number = 17411
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXA-GERM
Route/format = TOPICAL / EMULSION
Application Type = N
Applicant Name = HUNTINGTON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----
Product id = 14212
Application Number = 17412
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = GERMA-MEDICA
Route/format = TOPICAL / SOLUTION
Application Type = N
Applicant Name = HUNTINGTON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 1%
----
Product id = 14213
Application Number = 17412
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = GERMA-MEDICA "MG"
Route/format = TOPICAL / SOLUTION
Application Type = N
Applicant Name = HUNTINGTON LABS
ProductNo = 002
Tecode =
Rld = No
Strength = 0.25%
----
Product id = 14325
Application Number = 17413
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = SCRUBTEAM SURGICAL SPONGEBRUSH
Route/format = TOPICAL / SPONGE
Application Type = N
Applicant Name = 3M
ProductNo = 001
Tecode =
Rld = No
Strength = 330MG
----
Product id = 14159
Application Number = 17421
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DIAL
Route/format = TOPICAL / SOLUTION
Application Type = N
Applicant Name = DIAL
ProductNo = 002
Tecode =
Rld = No
Strength = 0.25%
----
Product id = 14276
Application Number = 17423
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = SEPTISOL
Route/format = TOPICAL / SOLUTION
Application Type = N
Applicant Name = VESTAL LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25%
----
Product id = 104
Application Number = 17424
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = SEPTISOL
Route/format = TOPICAL / AEROSOL
Application Type = N
Applicant Name = VESTAL LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 0.23%
----
Product id = 14323
Application Number = 17433
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = PRE-OP
Route/format = TOPICAL / SPONGE
Application Type = N
Applicant Name = DAVIS AND GECK
ProductNo = 001
Tecode = AT
Rld = Yes
Strength = 480MG
----
Product id = 14324
Application Number = 17433
Date of Application = Prior, Approved, to
RX/OTC/DISCN = RX
Tradename = PRE-OP II
Route/format = TOPICAL / SPONGE
Application Type = N
Applicant Name = DAVIS AND GECK
ProductNo = 002
Tecode = AT
Rld = No
Strength = 480MG
----
Product id = 14322
Application Number = 17446
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = PHISO-SCRUB
Route/format = TOPICAL / SPONGE
Application Type = N
Applicant Name = SANOFI AVENTIS US
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----
Product id = 14315
Application Number = 17452
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = E-Z SCRUB
Route/format = TOPICAL / SPONGE
Application Type = N
Applicant Name = BECTON DICKINSON
ProductNo = 001
Tecode =
Rld = No
Strength = 450MG
----
Product id = 14275
Application Number = 17460
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = SEPTI-SOFT
Route/format = TOPICAL / SOLUTION
Application Type = N
Applicant Name = CALGON
ProductNo = 001
Tecode =
Rld = No
Strength = 0.25%
----
Product id = 14318
Application Number = 18363
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = HEXASCRUB
Route/format = TOPICAL / SPONGE
Application Type = N
Applicant Name = PROF DSPLS
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----
Product id = 105
Application Number = 18375
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = TURGEX
Route/format = TOPICAL / AEROSOL
Application Type = N
Applicant Name = XTTRIUM
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----
Product id = 4599
Application Number = 19055
Date of Application = 30,, Nov, 1984
RX/OTC/DISCN = DISCN
Tradename = TURGEX
Route/format = TOPICAL / EMULSION
Application Type = N
Applicant Name = XTTRIUM
ProductNo = 001
Tecode =
Rld = No
Strength = 3%
----