ibutilide-fumarate


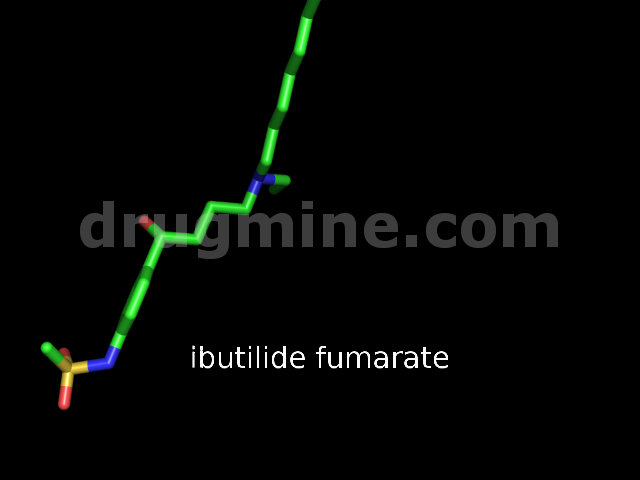
Name: IBUTILIDE FUMARATE
ID :
MW: 385
Number of atoms: 26
Molecular_Formula: C20H36N2O3S
Alogp: 3.992
Indication class : Cardiac Depressant (anti-arrhythmic)
Oral Flag : 0
Max_Phase : 4
Molecule_Type : Small molecule
ibutilide fumarate containing products summary
There are in total 4 different products containing the active ingredient ibutilide fumarate. From the 4 drug products, zero have been discontinued.Product id = 6861
Application Number = 20491
Date of Application = 28,, Dec, 1995
RX/OTC/DISCN = RX
Tradename = CORVERT
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = PHARMACIA AND UPJOHN
ProductNo = 001
Tecode = AP
Rld = Yes
Strength = 0.1MG/ML
----
Product id = 8357
Application Number = 90240
Date of Application = 11,, Jan, 2010
RX/OTC/DISCN = RX
Tradename = IBUTILIDE FUMARATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = LUITPOLD
ProductNo = 001
Tecode = AP
Rld = No
Strength = 0.1MG/ML
----
Product id = 8358
Application Number = 90643
Date of Application = 11,, Jan, 2010
RX/OTC/DISCN = RX
Tradename = IBUTILIDE FUMARATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = MYLAN INSTITUTIONAL
ProductNo = 001
Tecode = AP
Rld = No
Strength = 0.1MG/ML
----
Product id = 8359
Application Number = 90924
Date of Application = 11,, Jan, 2010
RX/OTC/DISCN = RX
Tradename = IBUTILIDE FUMARATE
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = MYLAN INSTITUTIONAL
ProductNo = 001
Tecode = AP
Rld = No
Strength = 0.1MG/ML
----