iopamidol
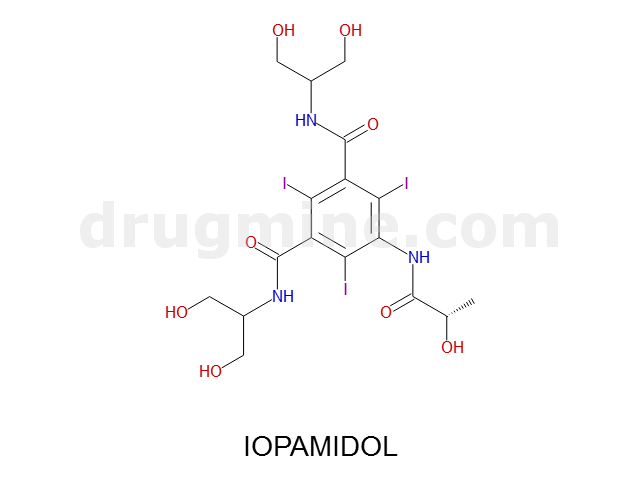
Name: IOPAMIDOL
ID :
MW: 777
Number of atoms: 31
Molecular_Formula: C17H22I3N3O8
Alogp: -1.187
Indication class : Diagnostic Aid (radiopaque medium)
Oral Flag : 0
Max_Phase : 4
Molecule_Type : Small molecule
iopamidol containing products summary
There are in total 39 different products containing the active ingredient iopamidol. From the 39 drug products, 28 have been discontinued.Product id = 8549
Application Number = 18735
Date of Application = 31,, Dec, 1985
RX/OTC/DISCN = RX
Tradename = ISOVUE-M 200
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 001
Tecode =
Rld = Yes
Strength = 41%
----
Product id = 8545
Application Number = 18735
Date of Application = 31,, Dec, 1985
RX/OTC/DISCN = RX
Tradename = ISOVUE-300
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 002
Tecode = AP
Rld = Yes
Strength = 61%
----
Product id = 8547
Application Number = 18735
Date of Application = 31,, Dec, 1985
RX/OTC/DISCN = RX
Tradename = ISOVUE-370
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 003
Tecode = AP
Rld = Yes
Strength = 76%
----
Product id = 8550
Application Number = 18735
Date of Application = 31,, Dec, 1985
RX/OTC/DISCN = RX
Tradename = ISOVUE-M 300
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 004
Tecode =
Rld = Yes
Strength = 61%
----
Product id = 8540
Application Number = 18735
Date of Application = 21,, Oct, 1986
RX/OTC/DISCN = DISCN
Tradename = ISOVUE-128
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 005
Tecode =
Rld = No
Strength = 26%
----
Product id = 8541
Application Number = 18735
Date of Application = 7,, Jul, 1987
RX/OTC/DISCN = RX
Tradename = ISOVUE-200
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 006
Tecode =
Rld = Yes
Strength = 41%
----
Product id = 8543
Application Number = 18735
Date of Application = 6,, Jul, 1992
RX/OTC/DISCN = RX
Tradename = ISOVUE-250
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 007
Tecode =
Rld = Yes
Strength = 51%
----
Product id = 8542
Application Number = 20327
Date of Application = 12,, Oct, 1994
RX/OTC/DISCN = DISCN
Tradename = ISOVUE-200
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 001
Tecode =
Rld = No
Strength = 41%
----
Product id = 8544
Application Number = 20327
Date of Application = 12,, Oct, 1994
RX/OTC/DISCN = RX
Tradename = ISOVUE-250
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 002
Tecode =
Rld = Yes
Strength = 51%
----
Product id = 8546
Application Number = 20327
Date of Application = 12,, Oct, 1994
RX/OTC/DISCN = RX
Tradename = ISOVUE-300
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 003
Tecode =
Rld = Yes
Strength = 61%
----
Product id = 8548
Application Number = 20327
Date of Application = 12,, Oct, 1994
RX/OTC/DISCN = RX
Tradename = ISOVUE-370
Route/format = INJECTION / INJECTABLE
Application Type = N
Applicant Name = BRACCO
ProductNo = 004
Tecode =
Rld = Yes
Strength = 76%
----
Product id = 8433
Application Number = 74629
Date of Application = 6,, Nov, 1996
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = BAXTER HLTHCARE
ProductNo = 001
Tecode =
Rld = No
Strength = 41%
----
Product id = 8435
Application Number = 74629
Date of Application = 6,, Nov, 1996
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = BAXTER HLTHCARE
ProductNo = 002
Tecode =
Rld = No
Strength = 61%
----
Product id = 8436
Application Number = 74629
Date of Application = 6,, Nov, 1996
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = BAXTER HLTHCARE
ProductNo = 003
Tecode =
Rld = No
Strength = 76%
----
Product id = 8434
Application Number = 74629
Date of Application = 31,, Mar, 1998
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = BAXTER HLTHCARE
ProductNo = 004
Tecode =
Rld = No
Strength = 51%
----
Product id = 8441
Application Number = 74636
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-200 IN PLASTIC CONTAINER
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 41%
----
Product id = 8446
Application Number = 74636
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-250 IN PLASTIC CONTAINER
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 002
Tecode =
Rld = No
Strength = 51%
----
Product id = 8452
Application Number = 74636
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300 IN PLASTIC CONTAINER
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 003
Tecode =
Rld = No
Strength = 61%
----
Product id = 8458
Application Number = 74636
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-370 IN PLASTIC CONTAINER
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 004
Tecode =
Rld = No
Strength = 76%
----
Product id = 8453
Application Number = 74637
Date of Application = 3,, Apr, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300 IN PLASTIC CONTAINER
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 61%
----
Product id = 8447
Application Number = 74638
Date of Application = 30,, Apr, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = ABBVIE
ProductNo = 001
Tecode =
Rld = No
Strength = 61%
----
Product id = 8443
Application Number = 74679
Date of Application = 2,, Apr, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-250
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = FRESENIUS KABI USA
ProductNo = 001
Tecode =
Rld = No
Strength = 51%
----
Product id = 8449
Application Number = 74679
Date of Application = 2,, Apr, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = FRESENIUS KABI USA
ProductNo = 002
Tecode =
Rld = No
Strength = 61%
----
Product id = 8455
Application Number = 74679
Date of Application = 2,, Apr, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-370
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = FRESENIUS KABI USA
ProductNo = 003
Tecode =
Rld = No
Strength = 76%
----
Product id = 8437
Application Number = 74734
Date of Application = 10,, Dec, 1996
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 61%
----
Product id = 8438
Application Number = 74734
Date of Application = 10,, Dec, 1996
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 002
Tecode =
Rld = No
Strength = 76%
----
Product id = 8439
Application Number = 74881
Date of Application = 28,, Jul, 2000
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-200
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = COOK IMAGING
ProductNo = 001
Tecode =
Rld = No
Strength = 41%
----
Product id = 8442
Application Number = 74881
Date of Application = 28,, Jul, 2000
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-250
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = COOK IMAGING
ProductNo = 002
Tecode =
Rld = No
Strength = 51%
----
Product id = 8448
Application Number = 74881
Date of Application = 28,, Jul, 2000
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = COOK IMAGING
ProductNo = 003
Tecode =
Rld = No
Strength = 61%
----
Product id = 8454
Application Number = 74881
Date of Application = 28,, Jul, 2000
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-370
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = COOK IMAGING
ProductNo = 004
Tecode =
Rld = No
Strength = 76%
----
Product id = 8440
Application Number = 74898
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-200
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 41%
----
Product id = 8444
Application Number = 74898
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-250
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 002
Tecode =
Rld = No
Strength = 51%
----
Product id = 8450
Application Number = 74898
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 003
Tecode =
Rld = No
Strength = 61%
----
Product id = 8456
Application Number = 74898
Date of Application = 30,, Dec, 1997
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-370
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 004
Tecode =
Rld = No
Strength = 76%
----
Product id = 8445
Application Number = 75005
Date of Application = 24,, Feb, 1998
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-250
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 001
Tecode =
Rld = No
Strength = 51%
----
Product id = 8451
Application Number = 75005
Date of Application = 24,, Feb, 1998
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-300
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 002
Tecode =
Rld = No
Strength = 61%
----
Product id = 8457
Application Number = 75005
Date of Application = 24,, Feb, 1998
RX/OTC/DISCN = DISCN
Tradename = IOPAMIDOL-370
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = HOSPIRA
ProductNo = 003
Tecode =
Rld = No
Strength = 76%
----
Product id = 10740
Application Number = 90394
Date of Application = 18,, Jun, 2010
RX/OTC/DISCN = RX
Tradename = SCANLUX-300
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = SANOCHEMIA CORP USA
ProductNo = 001
Tecode = AP
Rld = No
Strength = 61%
----
Product id = 10741
Application Number = 90394
Date of Application = 18,, Jun, 2010
RX/OTC/DISCN = RX
Tradename = SCANLUX-370
Route/format = INJECTION / INJECTABLE
Application Type = A
Applicant Name = SANOCHEMIA CORP USA
ProductNo = 002
Tecode = AP
Rld = No
Strength = 76%
----