loxapine-succinate
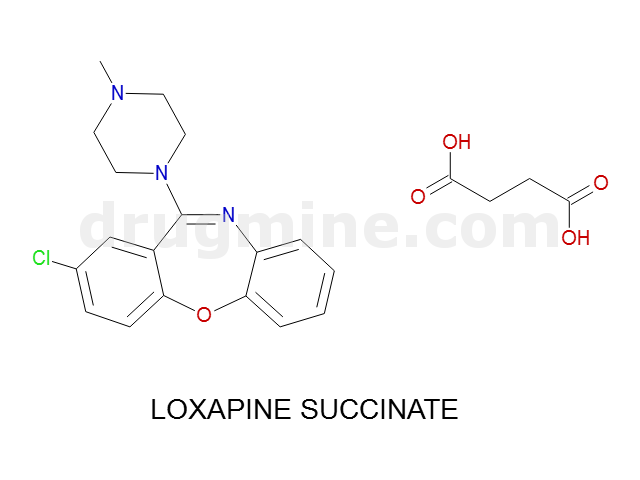
Name: LOXAPINE SUCCINATE
ID :
MW: 328
Number of atoms: 23
Molecular_Formula: C18H18ClN3O
Alogp: 3.433
Indication class : Tranquilizer (minor)
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
loxapine succinate containing products summary
There are in total 23 different products containing the active ingredient loxapine succinate. From the 23 drug products, 7 have been discontinued.Product id = 2426
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 5MG BASE
----
Product id = 2427
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 002
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 2428
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 003
Tecode =
Rld = No
Strength = EQ 25MG BASE
----
Product id = 2429
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 004
Tecode =
Rld = No
Strength = EQ 50MG BASE
----
Product id = 24080
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 006
Tecode =
Rld = No
Strength = EQ 10MG BASE
----
Product id = 24081
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 007
Tecode =
Rld = No
Strength = EQ 25MG BASE
----
Product id = 24082
Application Number = 17525
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = LOXITANE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 008
Tecode =
Rld = No
Strength = EQ 50MG BASE
----
Product id = 2425
Application Number = 72062
Date of Application = 15,, Jun, 1988
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 50MG BASE
----
Product id = 2422
Application Number = 72204
Date of Application = 15,, Jun, 1988
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 2423
Application Number = 72205
Date of Application = 15,, Jun, 1988
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 2424
Application Number = 72206
Date of Application = 15,, Jun, 1988
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = EQ 25MG BASE
----
Product id = 2418
Application Number = 76762
Date of Application = 1,, Nov, 2004
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 2419
Application Number = 76762
Date of Application = 1,, Nov, 2004
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 2420
Application Number = 76762
Date of Application = 1,, Nov, 2004
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 25MG BASE
----
Product id = 2421
Application Number = 76762
Date of Application = 1,, Nov, 2004
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 50MG BASE
----
Product id = 2414
Application Number = 76868
Date of Application = 4,, Aug, 2005
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MIKAH PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 2415
Application Number = 76868
Date of Application = 4,, Aug, 2005
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MIKAH PHARMA
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 2416
Application Number = 76868
Date of Application = 4,, Aug, 2005
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MIKAH PHARMA
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 25MG BASE
----
Product id = 2417
Application Number = 76868
Date of Application = 4,, Aug, 2005
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MIKAH PHARMA
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 50MG BASE
----
Product id = 2410
Application Number = 90695
Date of Application = 26,, Sep, 2011
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LANNETT HOLDINGS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = EQ 5MG BASE
----
Product id = 2411
Application Number = 90695
Date of Application = 26,, Sep, 2011
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LANNETT HOLDINGS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = EQ 10MG BASE
----
Product id = 2412
Application Number = 90695
Date of Application = 26,, Sep, 2011
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LANNETT HOLDINGS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = EQ 25MG BASE
----
Product id = 2413
Application Number = 90695
Date of Application = 26,, Sep, 2011
RX/OTC/DISCN = RX
Tradename = LOXAPINE SUCCINATE
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LANNETT HOLDINGS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = EQ 50MG BASE
----