modafinil

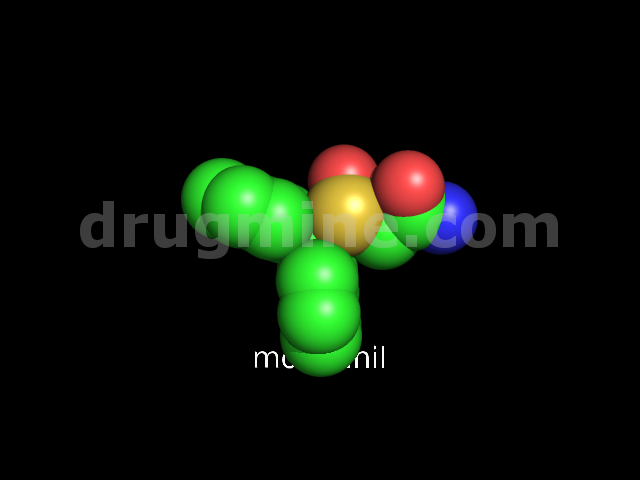

Name: MODAFINIL
ID :
MW: 273
Number of atoms: 19
Molecular_Formula: C15H15NO2S
Alogp: 2.05
Indication class : Analeptic (treatment of narcolepsy hypersomnia)
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
modafinil containing products summary
There are in total 16 different products containing the active ingredient modafinil. From the 16 drug products, zero have been discontinued.Product id = 27059
Application Number = 20717
Date of Application = 24,, Dec, 1998
RX/OTC/DISCN = RX
Tradename = PROVIGIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 27060
Application Number = 20717
Date of Application = 24,, Dec, 1998
RX/OTC/DISCN = RX
Tradename = PROVIGIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 002
Tecode = AB
Rld = Yes
Strength = 200MG
----
Product id = 25038
Application Number = 76594
Date of Application = 16,, Jul, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25039
Application Number = 76594
Date of Application = 16,, Jul, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25034
Application Number = 76715
Date of Application = 1,, Nov, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CARLSBAD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25035
Application Number = 76715
Date of Application = 1,, Nov, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CARLSBAD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25030
Application Number = 77667
Date of Application = 3,, Feb, 2014
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25031
Application Number = 77667
Date of Application = 3,, Feb, 2014
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25040
Application Number = 78963
Date of Application = 26,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25041
Application Number = 78963
Date of Application = 26,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25036
Application Number = 90543
Date of Application = 26,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HIKMA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25037
Application Number = 90543
Date of Application = 26,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HIKMA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25032
Application Number = 202566
Date of Application = 27,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25033
Application Number = 202566
Date of Application = 27,, Sep, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25028
Application Number = 202700
Date of Application = 18,, Oct, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ALEMBIC LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 100MG
----
Product id = 25029
Application Number = 202700
Date of Application = 18,, Oct, 2012
RX/OTC/DISCN = RX
Tradename = MODAFINIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ALEMBIC LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 200MG
----