mycophenolic-acid
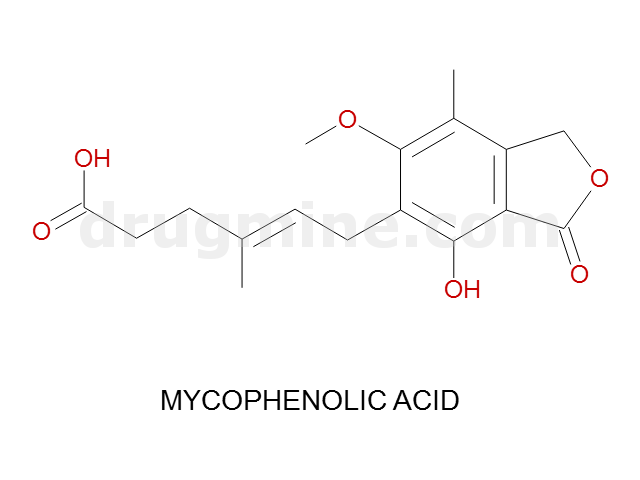
Name: MYCOPHENOLIC ACID
ID :
MW: 320
Number of atoms: 23
Molecular_Formula: C17H20O6
Alogp: 3.157
Indication class : Antineoplastic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
mycophenolic acid containing products summary
There are in total 8 different products containing the active ingredient mycophenolic acid. From the 8 drug products, zero have been discontinued.Product id = 15530
Application Number = 50791
Date of Application = 27,, Feb, 2004
RX/OTC/DISCN = RX
Tradename = MYFORTIC
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 180MG
----
Product id = 15531
Application Number = 50791
Date of Application = 27,, Feb, 2004
RX/OTC/DISCN = RX
Tradename = MYFORTIC
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = N
Applicant Name = NOVARTIS
ProductNo = 002
Tecode = AB
Rld = Yes
Strength = 360MG
----
Product id = 15527
Application Number = 91248
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = MYCOPHENOLIC ACID
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 360MG
----
Product id = 15526
Application Number = 91248
Date of Application = 8,, Jan, 2014
RX/OTC/DISCN = RX
Tradename = MYCOPHENOLIC ACID
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 180MG
----
Product id = 15524
Application Number = 91558
Date of Application = 21,, Aug, 2012
RX/OTC/DISCN = RX
Tradename = MYCOPHENOLIC ACID
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 180MG
----
Product id = 15525
Application Number = 91558
Date of Application = 19,, Aug, 2014
RX/OTC/DISCN = RX
Tradename = MYCOPHENOLIC ACID
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 360MG
----
Product id = 15528
Application Number = 202720
Date of Application = 30,, Oct, 2014
RX/OTC/DISCN = RX
Tradename = MYCOPHENOLIC ACID
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = A
Applicant Name = TEVA PHARMS USA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 180MG
----
Product id = 15529
Application Number = 202720
Date of Application = 30,, Oct, 2014
RX/OTC/DISCN = RX
Tradename = MYCOPHENOLIC ACID
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = A
Applicant Name = TEVA PHARMS USA
ProductNo = 002
Tecode = AB
Rld = No
Strength = 360MG
----