nevirapine
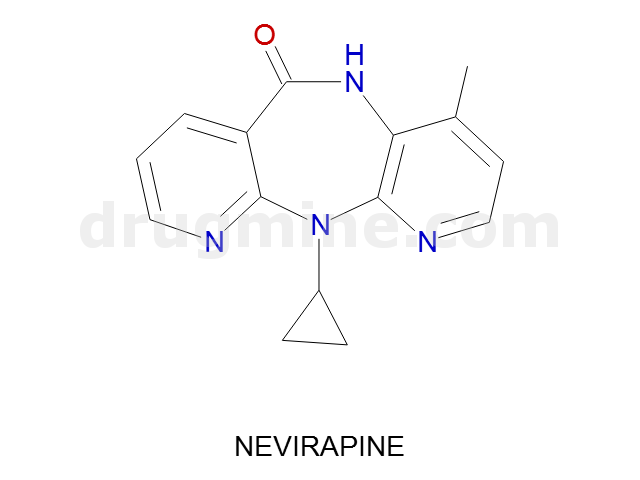

Name: NEVIRAPINE
ID :
MW: 266
Number of atoms: 20
Molecular_Formula: C15H14N4O
Alogp: 2.293
Indication class : Antiviral
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
nevirapine containing products summary
There are in total 18 different products containing the active ingredient nevirapine. From the 18 drug products, zero have been discontinued.Product id = 29595
Application Number = 20636
Date of Application = 21,, Jun, 1996
RX/OTC/DISCN = RX
Tradename = VIRAMUNE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = 200MG
----
Product id = 14837
Application Number = 20933
Date of Application = 11,, Sep, 1998
RX/OTC/DISCN = RX
Tradename = VIRAMUNE
Route/format = ORAL / SUSPENSION
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 001
Tecode = AA
Rld = Yes
Strength = 50MG/5ML
----
Product id = 25414
Application Number = 77521
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = AUROBINDO
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 14753
Application Number = 77702
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / SUSPENSION
Application Type = A
Applicant Name = AUROBINDO
ProductNo = 001
Tecode = AA
Rld = No
Strength = 50MG/5ML
----
Product id = 25415
Application Number = 77956
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = CIPLA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25422
Application Number = 78195
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = STRIDES PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25416
Application Number = 78584
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = HETERO LABS LTD III
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25420
Application Number = 78644
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = PRINSTON INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25418
Application Number = 78864
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 16772
Application Number = 201152
Date of Application = 25,, Mar, 2011
RX/OTC/DISCN = RX
Tradename = VIRAMUNE XR
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = 400MG
----
Product id = 16771
Application Number = 201152
Date of Application = 8,, Nov, 2012
RX/OTC/DISCN = RX
Tradename = VIRAMUNE XR
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = N
Applicant Name = BOEHRINGER INGELHEIM
ProductNo = 002
Tecode =
Rld = No
Strength = 100MG
----
Product id = 25419
Application Number = 202523
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25413
Application Number = 203021
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25417
Application Number = 203080
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MICRO LABS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 25421
Application Number = 203176
Date of Application = 22,, May, 2012
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SCIEGEN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 200MG
----
Product id = 16335
Application Number = 203411
Date of Application = 3,, Apr, 2014
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode = AB
Rld = No
Strength = 400MG
----
Product id = 16333
Application Number = 205258
Date of Application = 3,, Apr, 2014
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 400MG
----
Product id = 16334
Application Number = 205651
Date of Application = 27,, Oct, 2014
RX/OTC/DISCN = RX
Tradename = NEVIRAPINE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 400MG
----