oxybutynin-chloride
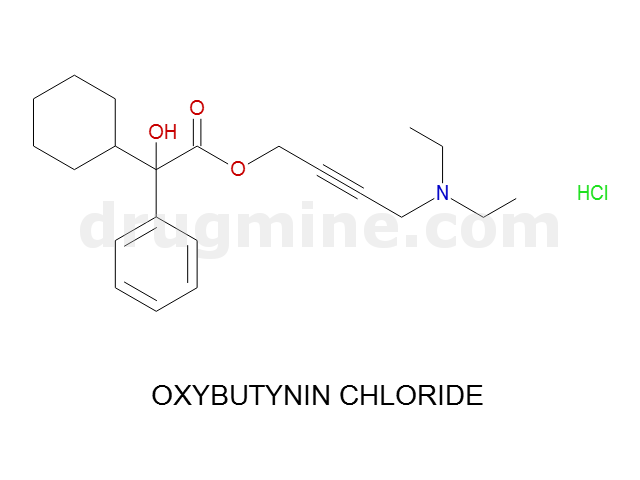
Name: OXYBUTYNIN CHLORIDE
ID :
MW: 357
Number of atoms: 26
Molecular_Formula: C22H31NO3
Alogp: 4.646
Indication class : Anticholinergic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
oxybutynin chloride containing products summary
There are in total 27 different products containing the active ingredient oxybutynin chloride. From the 27 drug products, 7 have been discontinued.Product id = 20568
Application Number = 17577
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DITROPAN
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = JANSSEN PHARMS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 14991
Application Number = 18211
Date of Application = Prior, Approved, to
RX/OTC/DISCN = DISCN
Tradename = DITROPAN
Route/format = ORAL / SYRUP
Application Type = N
Applicant Name = ORTHO MCNEIL JANSSEN
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 15877
Application Number = 20897
Date of Application = 16,, Dec, 1998
RX/OTC/DISCN = RX
Tradename = DITROPAN XL
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = N
Applicant Name = JANSSEN PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 15878
Application Number = 20897
Date of Application = 16,, Dec, 1998
RX/OTC/DISCN = RX
Tradename = DITROPAN XL
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = N
Applicant Name = JANSSEN PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 15879
Application Number = 20897
Date of Application = 22,, Jun, 1999
RX/OTC/DISCN = RX
Tradename = DITROPAN XL
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = N
Applicant Name = JANSSEN PHARMS
ProductNo = 003
Tecode = AB
Rld = Yes
Strength = 15MG
----
Product id = 5457
Application Number = 22204
Date of Application = 27,, Jan, 2009
RX/OTC/DISCN = RX
Tradename = GELNIQUE
Route/format = TRANSDERMAL / GEL
Application Type = N
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = Yes
Strength = 10%(100MG/PACKET)
----
Product id = 25747
Application Number = 70746
Date of Application = 10,, Mar, 1988
RX/OTC/DISCN = DISCN
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = USL PHARMA
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 25746
Application Number = 71655
Date of Application = 14,, Nov, 1988
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS USA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 25745
Application Number = 72296
Date of Application = 8,, Dec, 1988
RX/OTC/DISCN = DISCN
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = QUANTUM PHARMICS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 25750
Application Number = 72485
Date of Application = 19,, Apr, 1989
RX/OTC/DISCN = DISCN
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 15051
Application Number = 74520
Date of Application = 29,, Mar, 1996
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = SILARX
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 25748
Application Number = 74625
Date of Application = 31,, Jul, 1996
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = USL PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 15053
Application Number = 74868
Date of Application = 12,, Feb, 1997
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = WOCKHARDT
ProductNo = 001
Tecode = AA
Rld = Yes
Strength = 5MG/5ML
----
Product id = 15048
Application Number = 74997
Date of Application = 15,, Oct, 1997
RX/OTC/DISCN = DISCN
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 15049
Application Number = 75039
Date of Application = 29,, Jan, 1999
RX/OTC/DISCN = DISCN
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = MIKART
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG/5ML
----
Product id = 25749
Application Number = 75079
Date of Application = 31,, Oct, 1997
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = VINTAGE PHARMS
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = 5MG
----
Product id = 15050
Application Number = 75137
Date of Application = 18,, Dec, 1998
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = PHARM ASSOC
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 16427
Application Number = 76644
Date of Application = 9,, Nov, 2006
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 16428
Application Number = 76644
Date of Application = 10,, May, 2007
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 15MG
----
Product id = 15052
Application Number = 76682
Date of Application = 28,, Dec, 2004
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / SYRUP
Application Type = A
Applicant Name = VINTAGE PHARMS
ProductNo = 001
Tecode = AA
Rld = No
Strength = 5MG/5ML
----
Product id = 16426
Application Number = 76702
Date of Application = 9,, Nov, 2006
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 16425
Application Number = 76745
Date of Application = 9,, Nov, 2006
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = IMPAX PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 15MG
----
Product id = 16423
Application Number = 76745
Date of Application = 9,, May, 2007
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = IMPAX PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 16424
Application Number = 76745
Date of Application = 9,, May, 2007
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = IMPAX PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 16429
Application Number = 78503
Date of Application = 4,, Feb, 2009
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = OSMOTICA PHARM
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 16430
Application Number = 78503
Date of Application = 4,, Feb, 2009
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = OSMOTICA PHARM
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 16431
Application Number = 78503
Date of Application = 4,, Feb, 2009
RX/OTC/DISCN = RX
Tradename = OXYBUTYNIN CHLORIDE
Route/format = ORAL / TABLET, EXTENDED RELEASE
Application Type = A
Applicant Name = OSMOTICA PHARM
ProductNo = 003
Tecode = AB
Rld = No
Strength = 15MG
----