ramipril
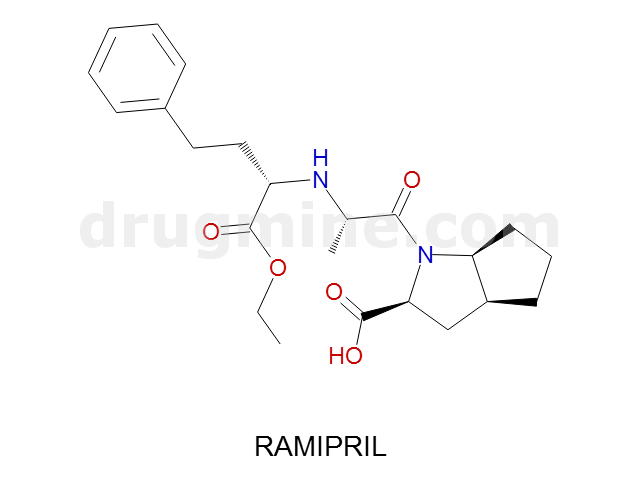
Name: RAMIPRIL
ID :
MW: 417
Number of atoms: 30
Molecular_Formula: C23H32N2O5
Alogp: 0.308
Indication class : Enzyme Inhibitor (angiotensin-converting); Antihypertensive
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
ramipril containing products summary
There are in total 70 different products containing the active ingredient ramipril. From the 70 drug products, 14 have been discontinued.Product id = 953
Application Number = 19901
Date of Application = 28,, Jan, 1991
RX/OTC/DISCN = RX
Tradename = ALTACE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = KING PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 954
Application Number = 19901
Date of Application = 28,, Jan, 1991
RX/OTC/DISCN = RX
Tradename = ALTACE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = KING PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 955
Application Number = 19901
Date of Application = 28,, Jan, 1991
RX/OTC/DISCN = RX
Tradename = ALTACE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = KING PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 956
Application Number = 19901
Date of Application = 28,, Jan, 1991
RX/OTC/DISCN = RX
Tradename = ALTACE
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = KING PHARMS
ProductNo = 004
Tecode = AB
Rld = Yes
Strength = 10MG
----
Product id = 17585
Application Number = 22021
Date of Application = 27,, Feb, 2007
RX/OTC/DISCN = DISCN
Tradename = ALTACE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = KING PFIZER
ProductNo = 001
Tecode =
Rld = No
Strength = 1.25MG
----
Product id = 17586
Application Number = 22021
Date of Application = 27,, Feb, 2007
RX/OTC/DISCN = DISCN
Tradename = ALTACE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = KING PFIZER
ProductNo = 002
Tecode =
Rld = No
Strength = 2.5MG
----
Product id = 17587
Application Number = 22021
Date of Application = 27,, Feb, 2007
RX/OTC/DISCN = DISCN
Tradename = ALTACE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = KING PFIZER
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 17588
Application Number = 22021
Date of Application = 27,, Feb, 2007
RX/OTC/DISCN = DISCN
Tradename = ALTACE
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = KING PFIZER
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 3142
Application Number = 76549
Date of Application = 24,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3143
Application Number = 76549
Date of Application = 24,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3144
Application Number = 76549
Date of Application = 24,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3145
Application Number = 76549
Date of Application = 24,, Oct, 2005
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = WATSON LABS
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3112
Application Number = 77004
Date of Application = 7,, Aug, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = CIPLA
ProductNo = 001
Tecode =
Rld = No
Strength = 1.25MG
----
Product id = 3113
Application Number = 77004
Date of Application = 7,, Aug, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = CIPLA
ProductNo = 002
Tecode =
Rld = No
Strength = 2.5MG
----
Product id = 3114
Application Number = 77004
Date of Application = 7,, Aug, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = CIPLA
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 3115
Application Number = 77004
Date of Application = 7,, Aug, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = CIPLA
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 3138
Application Number = 77470
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3139
Application Number = 77470
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3140
Application Number = 77470
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3141
Application Number = 77470
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3100
Application Number = 77513
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACTAVIS ELIZABETH
ProductNo = 001
Tecode =
Rld = No
Strength = 1.25MG
----
Product id = 3101
Application Number = 77513
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACTAVIS ELIZABETH
ProductNo = 002
Tecode =
Rld = No
Strength = 2.5MG
----
Product id = 3102
Application Number = 77513
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACTAVIS ELIZABETH
ProductNo = 003
Tecode =
Rld = No
Strength = 5MG
----
Product id = 3103
Application Number = 77513
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACTAVIS ELIZABETH
ProductNo = 004
Tecode =
Rld = No
Strength = 10MG
----
Product id = 3134
Application Number = 77514
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3135
Application Number = 77514
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3136
Application Number = 77514
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3137
Application Number = 77514
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3124
Application Number = 77626
Date of Application = 9,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3125
Application Number = 77626
Date of Application = 9,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3126
Application Number = 77626
Date of Application = 9,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3127
Application Number = 77626
Date of Application = 9,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = LUPIN
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3130
Application Number = 77900
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3131
Application Number = 77900
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ROXANE
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3132
Application Number = 77900
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ROXANE
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3133
Application Number = 77900
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ROXANE
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3116
Application Number = 78191
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3117
Application Number = 78191
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3118
Application Number = 78191
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3119
Application Number = 78191
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = DR REDDYS LABS LTD
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3120
Application Number = 78745
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3121
Application Number = 78745
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3122
Application Number = 78745
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3123
Application Number = 78745
Date of Application = 18,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = INVAGEN PHARMS
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3146
Application Number = 78832
Date of Application = 2,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3147
Application Number = 78832
Date of Application = 2,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3148
Application Number = 78832
Date of Application = 2,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3149
Application Number = 78832
Date of Application = 2,, Sep, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ZYDUS PHARMS USA
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3128
Application Number = 78849
Date of Application = 6,, Mar, 2009
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = RANBAXY LABS LTD
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 3129
Application Number = 78849
Date of Application = 6,, Mar, 2009
RX/OTC/DISCN = DISCN
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = RANBAXY LABS LTD
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 3104
Application Number = 79116
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3105
Application Number = 79116
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3106
Application Number = 79116
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3107
Application Number = 79116
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = APOTEX
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 27288
Application Number = 90650
Date of Application = 30,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 27289
Application Number = 90650
Date of Application = 30,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 27290
Application Number = 90650
Date of Application = 30,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 27291
Application Number = 90650
Date of Application = 30,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 27292
Application Number = 90697
Date of Application = 24,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 27293
Application Number = 90697
Date of Application = 24,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 27294
Application Number = 90697
Date of Application = 24,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 27295
Application Number = 90697
Date of Application = 24,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = ZYDUS PHARMS USA INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3108
Application Number = 91604
Date of Application = 8,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3109
Application Number = 91604
Date of Application = 8,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3110
Application Number = 91604
Date of Application = 8,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3111
Application Number = 91604
Date of Application = 8,, Jun, 2011
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA LTD
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3096
Application Number = 202392
Date of Application = 15,, Apr, 2014
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACCORD HLTHCARE INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 1.25MG
----
Product id = 3097
Application Number = 202392
Date of Application = 15,, Apr, 2014
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACCORD HLTHCARE INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 2.5MG
----
Product id = 3098
Application Number = 202392
Date of Application = 15,, Apr, 2014
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACCORD HLTHCARE INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3099
Application Number = 202392
Date of Application = 15,, Apr, 2014
RX/OTC/DISCN = RX
Tradename = RAMIPRIL
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ACCORD HLTHCARE INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = 10MG
----