riluzole
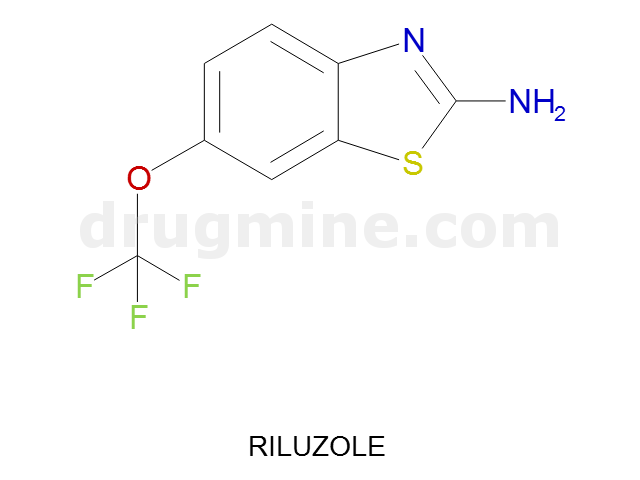


Name: RILUZOLE
ID :
MW: 234
Number of atoms: 15
Molecular_Formula: C8H5F3N2OS
Alogp: 3.828
Indication class : Amyotrophic Lateral Sclerosis Treatment
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
riluzole containing products summary
There are in total 6 different products containing the active ingredient riluzole. From the 6 drug products, zero have been discontinued.Product id = 27532
Application Number = 20599
Date of Application = 12,, Dec, 1995
RX/OTC/DISCN = RX
Tradename = RILUTEK
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = COVIS PHARMA SARL
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = 50MG
----
Product id = 27535
Application Number = 76173
Date of Application = 29,, Jan, 2003
RX/OTC/DISCN = RX
Tradename = RILUZOLE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = IMPAX LABS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 50MG
----
Product id = 27533
Application Number = 91300
Date of Application = 18,, Jun, 2013
RX/OTC/DISCN = RX
Tradename = RILUZOLE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX CORP
ProductNo = 001
Tecode = AB
Rld = No
Strength = 50MG
----
Product id = 27534
Application Number = 91394
Date of Application = 18,, Jun, 2013
RX/OTC/DISCN = RX
Tradename = RILUZOLE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = GLENMARK GENERICS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 50MG
----
Product id = 27537
Application Number = 91417
Date of Application = 18,, Jun, 2013
RX/OTC/DISCN = RX
Tradename = RILUZOLE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 50MG
----
Product id = 27536
Application Number = 203042
Date of Application = 1,, Jul, 2013
RX/OTC/DISCN = RX
Tradename = RILUZOLE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 50MG
----