risedronate-sodium
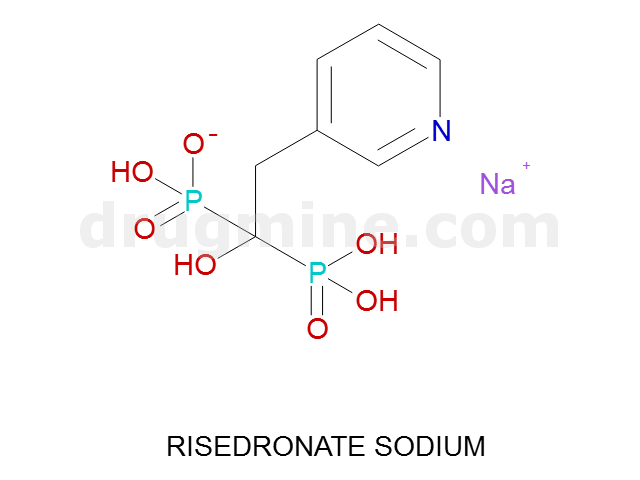


Name: RISEDRONATE SODIUM
ID :
MW: 282
Number of atoms: 17
Molecular_Formula: C7H10NO7P2
Alogp: -1.737
Indication class : Regulator (calcium)
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
risedronate sodium containing products summary
There are in total 17 different products containing the active ingredient risedronate sodium. From the 17 drug products, 2 have been discontinued.Product id = 17325
Application Number = 20835
Date of Application = 27,, Mar, 1998
RX/OTC/DISCN = RX
Tradename = ACTONEL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WARNER CHILCOTT LLC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 30MG
----
Product id = 17324
Application Number = 20835
Date of Application = 14,, Apr, 2000
RX/OTC/DISCN = RX
Tradename = ACTONEL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WARNER CHILCOTT LLC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 17326
Application Number = 20835
Date of Application = 25,, May, 2002
RX/OTC/DISCN = RX
Tradename = ACTONEL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WARNER CHILCOTT LLC
ProductNo = 003
Tecode = AB
Rld = Yes
Strength = 35MG
----
Product id = 17327
Application Number = 20835
Date of Application = 16,, Apr, 2007
RX/OTC/DISCN = DISCN
Tradename = ACTONEL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WARNER CHILCOTT LLC
ProductNo = 004
Tecode =
Rld = No
Strength = 75MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 17328
Application Number = 20835
Date of Application = 22,, Apr, 2008
RX/OTC/DISCN = RX
Tradename = ACTONEL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = WARNER CHILCOTT LLC
ProductNo = 005
Tecode = AB
Rld = Yes
Strength = 150MG
----
Product id = 17131
Application Number = 21823
Date of Application = 12,, Aug, 2005
RX/OTC/DISCN = DISCN
Tradename = ACTONEL WITH CALCIUM (COPACKAGED)
Route/format = ORAL / TABLET, TABLET
Application Type = N
Applicant Name = WARNER CHILCOTT
ProductNo = 001
Tecode =
Rld = No
Strength = EQ 500MG BASE,N/A;N/A,35MG **Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons**
----
Product id = 15399
Application Number = 22560
Date of Application = 8,, Oct, 2010
RX/OTC/DISCN = RX
Tradename = ATELVIA
Route/format = ORAL / TABLET, DELAYED RELEASE
Application Type = N
Applicant Name = WARNER CHILCOTT LLC
ProductNo = 001
Tecode =
Rld = Yes
Strength = 35MG
----
Product id = 27549
Application Number = 77132
Date of Application = 5,, Oct, 2007
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 27550
Application Number = 77132
Date of Application = 5,, Oct, 2007
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 30MG
----
Product id = 27551
Application Number = 77132
Date of Application = 5,, Oct, 2007
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 003
Tecode = AB
Rld = No
Strength = 35MG
----
Product id = 27552
Application Number = 79215
Date of Application = 13,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = TEVA PHARMS USA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 150MG
----
Product id = 27543
Application Number = 90877
Date of Application = 10,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 75MG
----
Product id = 27544
Application Number = 90877
Date of Application = 10,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = APOTEX INC
ProductNo = 003
Tecode = AB
Rld = No
Strength = 150MG
----
Product id = 27547
Application Number = 90886
Date of Application = 10,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARMA GLOBAL
ProductNo = 004
Tecode = AB
Rld = No
Strength = 75MG
----
Product id = 27548
Application Number = 90886
Date of Application = 10,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARMA GLOBAL
ProductNo = 005
Tecode = AB
Rld = No
Strength = 150MG
----
Product id = 27545
Application Number = 200477
Date of Application = 10,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 004
Tecode = AB
Rld = No
Strength = 75MG
----
Product id = 27546
Application Number = 200477
Date of Application = 10,, Jun, 2014
RX/OTC/DISCN = RX
Tradename = RISEDRONATE SODIUM
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = MYLAN PHARMS INC
ProductNo = 005
Tecode = AB
Rld = No
Strength = 150MG
----