tiagabine-hydrochloride
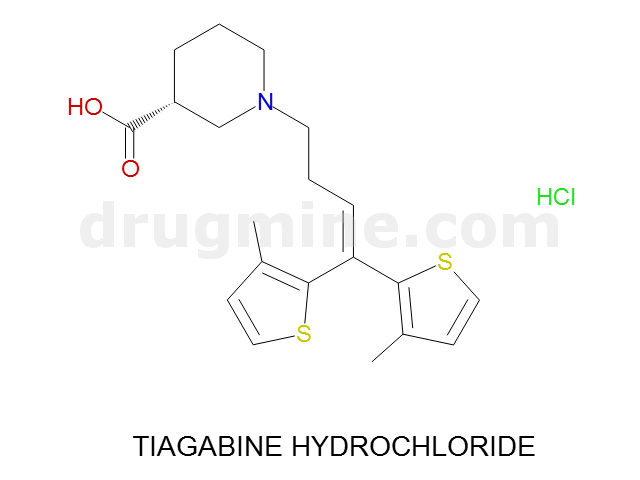


Name: TIAGABINE HYDROCHLORIDE
ID :
MW: 376
Number of atoms: 25
Molecular_Formula: C20H25NO2S2
Alogp: 2.252
Indication class : Anticonvulsant
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
tiagabine hydrochloride containing products summary
There are in total 10 different products containing the active ingredient tiagabine hydrochloride. From the 10 drug products, 4 have been discontinued.Product id = 21710
Application Number = 20646
Date of Application = 30,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 001
Tecode = AB
Rld = Yes
Strength = 4MG
----
Product id = 21714
Application Number = 20646
Date of Application = 30,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 002
Tecode =
Rld = No
Strength = 12MG
----
Product id = 21715
Application Number = 20646
Date of Application = 30,, Sep, 1997
RX/OTC/DISCN = RX
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 003
Tecode =
Rld = No
Strength = 16MG
----
Product id = 21716
Application Number = 20646
Date of Application = 30,, Sep, 1997
RX/OTC/DISCN = DISCN
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 004
Tecode =
Rld = No
Strength = 20MG
----
Product id = 21709
Application Number = 20646
Date of Application = 16,, Apr, 1999
RX/OTC/DISCN = RX
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 005
Tecode = AB
Rld = No
Strength = 2MG
----
Product id = 21711
Application Number = 20646
Date of Application = 29,, Nov, 2005
RX/OTC/DISCN = DISCN
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 006
Tecode =
Rld = No
Strength = 6MG
----
Product id = 21712
Application Number = 20646
Date of Application = 29,, Nov, 2005
RX/OTC/DISCN = DISCN
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 007
Tecode =
Rld = No
Strength = 8MG
----
Product id = 21713
Application Number = 20646
Date of Application = 29,, Nov, 2005
RX/OTC/DISCN = DISCN
Tradename = GABITRIL
Route/format = ORAL / TABLET
Application Type = N
Applicant Name = CEPHALON
ProductNo = 008
Tecode =
Rld = No
Strength = 10MG
----
Product id = 28703
Application Number = 77555
Date of Application = 4,, Nov, 2011
RX/OTC/DISCN = RX
Tradename = TIAGABINE HYDROCHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 2MG
----
Product id = 28704
Application Number = 77555
Date of Application = 4,, Nov, 2011
RX/OTC/DISCN = RX
Tradename = TIAGABINE HYDROCHLORIDE
Route/format = ORAL / TABLET
Application Type = A
Applicant Name = SUN PHARM INDS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 4MG
----