zaleplon
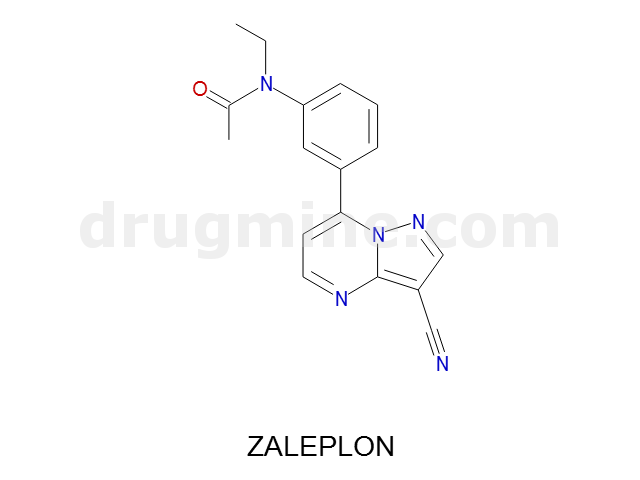

Name: ZALEPLON
ID :
MW: 305
Number of atoms: 23
Molecular_Formula: C17H15N5O
Alogp: 1.604
Indication class : Sedative-Hypnotic
Oral Flag : 1
Max_Phase : 4
Molecule_Type : Small molecule
zaleplon containing products summary
There are in total 22 different products containing the active ingredient zaleplon. From the 22 drug products, 2 have been discontinued.Product id = 3304
Application Number = 20859
Date of Application = 13,, Aug, 1999
RX/OTC/DISCN = RX
Tradename = SONATA
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = PFIZER
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3305
Application Number = 20859
Date of Application = 13,, Aug, 1999
RX/OTC/DISCN = RX
Tradename = SONATA
Route/format = ORAL / CAPSULE
Application Type = N
Applicant Name = PFIZER
ProductNo = 002
Tecode = AB
Rld = Yes
Strength = 10MG
----
Product id = 3784
Application Number = 77237
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ROXANE
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3785
Application Number = 77237
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ROXANE
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3780
Application Number = 77238
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3781
Application Number = 77238
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = MYLAN
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3788
Application Number = 77239
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3789
Application Number = 77239
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = TEVA PHARMS
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3776
Application Number = 77505
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3777
Application Number = 77505
Date of Application = 20,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = CIPLA LTD
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3786
Application Number = 78095
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 001
Tecode =
Rld = No
Strength = 5MG
----
Product id = 3787
Application Number = 78095
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = DISCN
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = SANDOZ
ProductNo = 002
Tecode =
Rld = No
Strength = 10MG
----
Product id = 3778
Application Number = 78147
Date of Application = 25,, Nov, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = HIKMA PHARMS LLC
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3779
Application Number = 78147
Date of Application = 25,, Nov, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = HIKMA PHARMS LLC
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3792
Application Number = 78706
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3793
Application Number = 78706
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = UPSHER SMITH
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3774
Application Number = 78829
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3775
Application Number = 78829
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = AUROBINDO PHARMA
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3790
Application Number = 78989
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = UNICHEM
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3791
Application Number = 78989
Date of Application = 6,, Jun, 2008
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = UNICHEM
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----
Product id = 3782
Application Number = 90374
Date of Application = 17,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 001
Tecode = AB
Rld = No
Strength = 5MG
----
Product id = 3783
Application Number = 90374
Date of Application = 17,, Sep, 2009
RX/OTC/DISCN = RX
Tradename = ZALEPLON
Route/format = ORAL / CAPSULE
Application Type = A
Applicant Name = ORCHID HLTHCARE
ProductNo = 002
Tecode = AB
Rld = No
Strength = 10MG
----